
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20, Problem 62P
What two monomers are needed to prepare each
a. 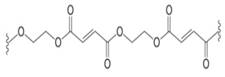 b.
b. 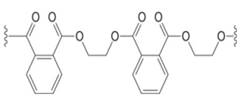
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Wacker's process involves:
Select one:
a. Hydrogenation of ethylene
O b. Hydroformylation process
c. Polymerization of ethylene
d. Oxidation of ethylene
69. The structure of the aldehyde or ketone that the
following compound came from:
H
A. III only
B. IV only
C. I and IV
D. II only
IV
draw the structures for: A. Acetic acid (Ethanoic acid) B. Benzoic acid
Chapter 20 Solutions
Organic Chemistry (6th Edition)
Ch. 20.1 - Prob. 1PCh. 20.2 - Draw the three possible resonance structures for...Ch. 20.2 - Prob. 3PCh. 20.3 - Give an IUPAC or common name for each compound. a....Ch. 20.3 - Problem 22.5 Draw the structure corresponding to...Ch. 20.4 - Problem 22.6 Explain why the boiling point of is...Ch. 20.7 - Prob. 11PCh. 20.7 - Prob. 12PCh. 20.8 - Prob. 13PCh. 20.9 - Problem 22.16 Draw the products of each reaction.
...
Ch. 20.9 - Prob. 17PCh. 20.9 - Problem 22.18 Draw a stepwise mechanism for the...Ch. 20.9 - Prob. 19PCh. 20.10 - Problem 22.20 Fenofibrate is a...Ch. 20.10 - Problem 22.21 What product is formed when the...Ch. 20 - Prob. 33PCh. 20 - 22.40 Give the IUPAC or common name for each...Ch. 20 - 22.41 Give the structure corresponding to each...Ch. 20 - Prob. 36PCh. 20 - 22.43 Explain why is a stronger acid and a weaker...Ch. 20 - (a) Propose an explanation for the difference in...Ch. 20 - Draw the product formed when phenylacetic acid is...Ch. 20 - Prob. 42PCh. 20 - Prob. 43PCh. 20 - Prob. 44PCh. 20 - Prob. 45PCh. 20 - Prob. 46PCh. 20 - Prob. 47PCh. 20 - Prob. 48PCh. 20 - 22.64 What carboxylic acid and alcohol are needed...Ch. 20 - Problem 22.65 Devise a synthesis of each compound...Ch. 20 - 22.70 What polyester or poly amide can be prepared...Ch. 20 - 22.71 What two monomers are needed to prepare each...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2. Differentiate acetic acid from hydrochloric acid in terms of. a. Boiling point b. Acidity c. Solubility in ethyl alcoholarrow_forwardDraw the structure of an alkane that: a. Contains only 1° and 4° carbons. c. Contains only 1° and 2° hydrogens. b. Contains only 2° carbons. d. Contains only 1° and 3° hydrogens.arrow_forward11. Caffeine is differentiated from theobromine: A. Easily soluble in hot water B. Soluble in acids C. Soluble in alkali solutions D. Easily soluble in chloroformarrow_forward
- The most common industrial application of diethyl phthalate (or more generally, phthalate esters) is as a plasticizer. What is a plasticizer? Explain why plastics become brittle over time.arrow_forwardGlycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forward49. Which of the follwing compounds is 4-iodo-6-cyclopropyl-3-hexanone? a. c. d. b.arrow_forward
- MCQ 22: Propan-2-ol will get oxidized to give A. aldehyde B. ketone C. alcohol D. carbonylarrow_forwardName : R NH2 Select one: a. alcohols b. amine c. phenols d. aldehydearrow_forwardAn acid catalyst in nucleophilic addition of aldehydes and ketones is used for: Select one: a. Protonation of carbonyl carbon b. Making the aldehyde and ketone more susceptible to nucleophiles c. Increasing the nucleophilicity of the nucleophile d. To provide a medium for the reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY