
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20, Problem 57P
Devise a synthesis of each compound using 1-bromobutane
a. 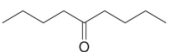 b.
b. 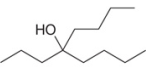
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the product(s) formed when A is treated with each reagent.a. NaBH4, CH3OH b. LiAlH4, then H2O c. Ag2O, NH4OH d. CrO3, H2SO4, H2O e. PCC
Devise a synthesis of each compound using 1-bromobutane (CH3CH2CH2CH2Br) as the only organic starting material. You may use any other inorganic reagents.
Devise a synthesis of each compound from benzene. You may use
alcohols with one or two carbons and any inorganic reagents.
a.
qe
Br
H
Br
b.
C.
N
OH
Chapter 20 Solutions
Organic Chemistry (6th Edition)
Ch. 20.1 - Prob. 1PCh. 20.2 - Draw the three possible resonance structures for...Ch. 20.2 - Prob. 3PCh. 20.3 - Give an IUPAC or common name for each compound. a....Ch. 20.3 - Problem 22.5 Draw the structure corresponding to...Ch. 20.4 - Problem 22.6 Explain why the boiling point of is...Ch. 20.7 - Prob. 11PCh. 20.7 - Prob. 12PCh. 20.8 - Prob. 13PCh. 20.9 - Problem 22.16 Draw the products of each reaction.
...
Ch. 20.9 - Prob. 17PCh. 20.9 - Problem 22.18 Draw a stepwise mechanism for the...Ch. 20.9 - Prob. 19PCh. 20.10 - Problem 22.20 Fenofibrate is a...Ch. 20.10 - Problem 22.21 What product is formed when the...Ch. 20 - Prob. 33PCh. 20 - 22.40 Give the IUPAC or common name for each...Ch. 20 - 22.41 Give the structure corresponding to each...Ch. 20 - Prob. 36PCh. 20 - 22.43 Explain why is a stronger acid and a weaker...Ch. 20 - (a) Propose an explanation for the difference in...Ch. 20 - Draw the product formed when phenylacetic acid is...Ch. 20 - Prob. 42PCh. 20 - Prob. 43PCh. 20 - Prob. 44PCh. 20 - Prob. 45PCh. 20 - Prob. 46PCh. 20 - Prob. 47PCh. 20 - Prob. 48PCh. 20 - 22.64 What carboxylic acid and alcohol are needed...Ch. 20 - Problem 22.65 Devise a synthesis of each compound...Ch. 20 - 22.70 What polyester or poly amide can be prepared...Ch. 20 - 22.71 What two monomers are needed to prepare each...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Design a synthesis of each compound from alcohols having four or fewer carbons, acetylene, and ethylene oxide as the only organic starting materials. You may use any other inorganic reagents you choose. a. b. C. CO₂H xx но d. OHarrow_forwardDevise a synthesis of each compound from benzene. You may also use any organic compounds having four carbons or fewer, and any required inorganic reagents. Co,CH3 a. b.arrow_forward16.69 Use the reactions in this chapter along with those learned in Chapters 11 and 12 to synthesize each compaund. You may use benzene, acetylene (HC=CH), ethanol, ethylene oxide, and any inorganic reagents. a. b. CI он C ON d. NO2arrow_forward
- 1. Draw the products of each nucleophilic substitution reaction a. b. D b OH C. d. e. f. Br 1 NaCN + NaOCH3 H₂Oarrow_forwardDesign a synthesis of each compound from alcohols having four or fewer carbons, acetylene, and ethylene oxide as the only organic starting materials. You may use any other inorganic reagents you choose. a. b. C. CO₂Harrow_forwardSynthesize each compound from cyclohexanol using any other organic or inorganic compounds. CH,OH a. g. (Each cyclohexane ring must come from cyclohexanol.) COOH b. d. h. сно CHs (Each cyclohexane ring must come from cyclohexanol.)arrow_forward
- Draw the product formed when each compound is treated with two equivalents of CH;CH,CH,CHgMgBr followed by H,0. OCH3 a. b. C. CH;CH2 OCH2CH3arrow_forwardCompound that is most easily hydrolyzed by acid in water `NH A. В. F C. D.arrow_forwardDevise a synthesis of each compound. You may use HC=CH, ethylene oxide, and alkyl halides as organic starting materials and any inorganic reagents.arrow_forward
- Draw a tautomer of each compound. NH a. b. C. .C. CH, NH2 NHCH CH;CH HO.arrow_forwardDevise a synthesis of each compound from an alkyl halide using any other organic or inorganic reagents. SH b. c. CH;CH,CN d. e. CH,CH,OCOCH3 a.arrow_forwardInstructions: Give the IUPAC name for each thiol. a. b. CH3CH2CHCH2CH3 SH SH c. (CH3CH₂)2CHCH₂CH₂CH₂CH₂SHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY