
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 38P
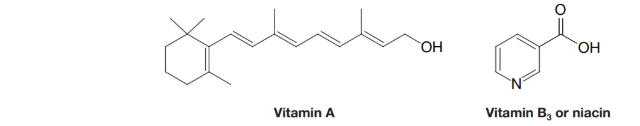
(a) Indicate the hydrophobic and hydrophilic parts of vitamin A and comment on whether you would expect it to be soluble in water. (b) Do the same for vitamin
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Palmitoleic acid, a fatty acid with various pharmaceutical
applications, is mainly obtained from macadamia nuts. The
condensed structural formula for a triacylglycerol containing
three palmitoleic acid units is provided below. Which of the
following statements is NOT true regarding this
triacylglycerol?
O
||
CH,−O−C–(CH,)–CH=CH—(CH2)5–CH3
O
||
CH−O−C−(CH2)–CH=CH–(CH,)5–CH3
O
CH,−O−C−(CH,)–CH=CH–(CH,)5–CH,
O
Its name is glyceryl tripalmitate or tripalmitin.
O It is most likely to be liquid at room temperature.
O It is an oil (not a fat).
O It contains 3 molecules of the same unsaturated fatty acid.
Explain why cholesterol, a compound with molecular formula C 27H 46O and one OH group, is soluble in CCl 4 but insoluble in water.
Complete the following reactions and hence identify the nature of the acid-base
interaction and the name associated with acid-base interaction that you have chosen.
Q1.
(a)
H30* + OH >
Chapter 2 Solutions
Organic Chemistry
Ch. 2 - Practice Problem 2.1
Propose structures for two...Ch. 2 - Prob. 2PPCh. 2 - Prob. 3PPCh. 2 - Prob. 4PPCh. 2 - Prob. 5PPCh. 2 - Practice Problem 2.6
Using a three-dimensional...Ch. 2 - Practice Problem 2.7
Trichloromethane (, also...Ch. 2 - Prob. 8PPCh. 2 - Prob. 9PPCh. 2 - Practice Problem 2.10
Write bond-line structural...
Ch. 2 - Practice Problem 2.11 Although we shall discuss...Ch. 2 - Practice Problem 2.12 Write bond-line structural...Ch. 2 - Prob. 13PPCh. 2 - Practice Problem 2.14
One way of naming ethers is...Ch. 2 - Practice Problem 2.15 Eugenol is the main...Ch. 2 - Practice Problem 2.16
One way of naming amines is...Ch. 2 - Practice Problem 2.17 Which amines in Practice...Ch. 2 - Prob. 18PPCh. 2 - Prob. 19PPCh. 2 - Practice Problem 2.20
Write bond-line formulas for...Ch. 2 - Practice Problem 2.21
Write bond-line formulas for...Ch. 2 - Practice Problem 2.22
Write bond-line formulas for...Ch. 2 - Prob. 23PPCh. 2 - Practice Problem 2.24 Write another resonance...Ch. 2 - Prob. 25PPCh. 2 - Practice Problem 2.26
Which compound would you...Ch. 2 - Practice Problem 2.27 Arrange the following...Ch. 2 - Prob. 28PPCh. 2 - Prob. 29PCh. 2 - Identify all of the functional groups in each of...Ch. 2 - 2.31 There are four alkyl bromides with the...Ch. 2 - Prob. 32PCh. 2 - Classify the following alcohols as primary,...Ch. 2 - 2.34 Classify the following amines as primary,...Ch. 2 - Prob. 35PCh. 2 - Identify all of the functional groups in Crixivan,...Ch. 2 - 2.37 Identify all of the functional groups in...Ch. 2 - 2.38 (a) Indicate the hydrophobic and hydrophilic...Ch. 2 - Hydrogen fluoride has a dipole moment of 1.83 D;...Ch. 2 - 2.40 Why does one expect the cis isomer of an...Ch. 2 - Prob. 41PCh. 2 - Prob. 42PCh. 2 - Prob. 43PCh. 2 - 2.44 Consider each of the following molecules in...Ch. 2 - True or false: For a molecule to be polar, the...Ch. 2 - 2.46 Which compound in each of the following...Ch. 2 - Prob. 47PCh. 2 - The IR spectrum of propanoic acid (Fig. 2.16)...Ch. 2 - Prob. 49PCh. 2 - Write structural formulas for four compounds with...Ch. 2 - There are four amides with the formula C3H7NO. (a)...Ch. 2 - Prob. 52PCh. 2 - Prob. 53PCh. 2 - Prob. 54PCh. 2 - Prob. 55PCh. 2 - 2.56 Compound C is asymmetric, has molecular...Ch. 2 - 2.57 Examine the diagram showing an -helical...Ch. 2 - Prob. 1LGPCh. 2 - Prob. 2LGPCh. 2 - Prob. 3LGPCh. 2 - Consider the molecular formula C4H8O2. Predict...Ch. 2 - Consider the molecular formula C4H8O2. If any of...Ch. 2 - Prob. 6LGPCh. 2 - Consider the molecular formula.
7. Pick five...Ch. 2 - Prob. 8LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
A liquid mixture contains 60.0 wt% ethanol (E), 5.0 wt% of a dissolved solute (S), and the balance water. A str...
Elementary Principles of Chemical Processes, Binder Ready Version
Q4. Which property of rubbing alcohol is a chemical property?
a) Density (0.786 g/cm3)
b) Flammability
c) Bo...
Chemistry: A Molecular Approach (4th Edition)
For each of the following molecules, (1) draw the correct Lewis structure; (2) label each polar covalent bond w...
General, Organic, and Biological Chemistry (3rd Edition)
9.1 Calculate the total mass of the reactants and the products for each of the following equations:
Basic Chemistry (5th Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
1.2 Ask two of your friends (not in class) to define the terms in problem1.1.
Do their answers agee with the d...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- During the hydrolysis of proteins, some amino acids, such as tryptophan, do not survive the reaction conditions. Other amino acids, such as asparagine and glutamine, are modified. Referring to Table 1-7 (p. 39), which shows the structures of the 20 common amino acids, write the structures of the two amino acids that are formed when asparagine and glutamine decompose in hot, concentrated HCl.arrow_forward(a) Which of the common amino acids have more than one carboxyl group?arrow_forwardIndicate whether each statement is true or false: (a) Fat molecules contain amide bonds. (b) Phosphoplipids can be zwitterions. (c) Phospholipids form bilayers in water in order to have their long hydrophobic tails interact favorably with each other, leaving their polar heads to the aqueous environment.arrow_forward
- Classify each molecule as hydrophilic, hydrophobic, or amphipathic (amphiphilic).arrow_forward10) The a helix of the secondary structure of a protein is held together by hydrogen bonds between two different parts of a protein chain. True or False 1) Hydrophobic interactions help to stabilize the tertiary and quaternary structures of a protein. True or Falsearrow_forwardQ1. The pKa values of Glutamic acid are 2.2, 4.3 and 9.7. H2N HO. OH (a) Calculate the pH of a glutamic acid solution in which the a-amine group is 80% deprotonated? (show your calculation!) (b) Draw the ionic forms of glutamic acid that exist at the calculated pH.arrow_forward
- Arrange the following acids in the increasing order of their solubility in water: p-ClC6H4COOH, CH3COOH, CH3(CH2)3COOH. Explain why their order became like that?arrow_forward(b) Polymers play essential and ubiquitous roles in our everyday life because of their broad spectrum of properties. (i) List TWO (2) polymers that you encounter everyday. (ii) Describe the properties of each polymer in (i).arrow_forwardclassify the molecules of interest (i.e. para-aminophenol, acetaminophen, phenacetin) in increasing order of polarity? Rationalize these observations based on molecular interactions.arrow_forward
- (i) Give two examples of macromolecules that are chosen as drug targets.(ii) What are antiseptics? Give an example.(iii) Why is use of aspartame limited to cold foods and soft drinks?arrow_forwardIntramolecular forces of attraction are often important in holding large molecules together. For example, some proteins fold into compact shapes, held together by attractive forces between nearby functional groups. A schematic of a folded protein is drawn here, with the protein backbone indicated by a blue-green ribbon, and various appendages drawn dangling from the chain. What types of intramolecular forces occur at each labeled site (A–F)?arrow_forwardExplain the difference in the solubility of benzoic acid and sodium benzoate in water.Which of the two would you predict to be more soluble in CHCl3? Explain.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY