
Concept explainers
Practice Problem 2.11
Although we shall discuss the naming of organic compounds later when we discuss the individual families in detail, one method of naming
What are the names for (c) 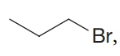 , (d)
, (d) 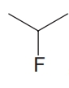 , and (e)
, and (e)
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Essential Organic Chemistry (3rd Edition)
Chemistry: A Molecular Approach (4th Edition)
Organic Chemistry
General Chemistry: Atoms First
Elementary Principles of Chemical Processes, Binder Ready Version
Principles of Chemistry: A Molecular Approach (3rd Edition)
- (Organic Chemistry) Name the following compounds:arrow_forward3.39 (a) Which of the following molecules can hydrogen bond to another molecule like itself? (b) Which of the following molecules eán hydrogen bond to water? OH СНО NH N. Aarrow_forward1. (a) Write the IUPAC name for each of the following compounds. (i) HO, CI ÓH (ii) Br (ii)arrow_forward
- Problem Name the ionic compound formed from the following pairs of elements: (a) magnesium and nitrogen; (b) iodine and cadmium; (c) strontium and fluorine; (d) sulfur and cesium.Plan The key to naming a binary ionic compound is to recognize which element is the metal and which is the nonmetal. When in doubt, check the periodic table. We place the cation name first, add the suffix -ide to the nonmetal root, and place the anion name last.arrow_forwardGive a clear handwritten answer with explanation.....give the name of given molecule..please give name all compoundsarrow_forwardGive a definition and an example for each class of organic compounds.(a) alkane (b) alkene (c) alkyne(d) alcohol (e) ether (f) ketonearrow_forward
- Directions: ONLY HANDWRITE YOUR ANSWERS IN THE SPACE PROVIDED and upload all pages as ONE unit as a pdf in Modules on Canvas. (Please note that some of these compounds do not exist as a stable substance in nature, however the following should serve as a practice exercise in nomenclature.) If provided the name, then you must write the formula. If provided the formula, then you must write the correct name. Indicate IUPAC names only. Use one of the following descriptions: IONIC, COVALENT, ACID or HYDRATE. 198. stannic oxalate 199. Sn(HCO3)4 200. CuC2O4arrow_forwardName the following compounds! a) b) c) d) archarrow_forwardWrite the name of the compounds according to IUPAC.arrow_forward
- You have an acyclic alkane hydrocarbon that has a total of 17 atoms. Which elements are in the formula? What is the molecular (chemical) formula? E.C: If the 17 atoms represents an UNSATURATED hydrocarbon, what is a possible formula?arrow_forwardName the following compounds. I just need part Z, AA, and BB.arrow_forwardname the following molecules with IUPAC and do IUPAC drawing as well. Black: Carbon White: Hydrogen Red: Oxygen Green: Chlorine Yellow: Sulfur Tan: Iodinearrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

