
Concept explainers
(a)
Interpretation:
Lewis structure, VSEPR formula, bond angle, and molecular shape for
Concept Introduction:
Valence Shell Electron Pair Repulsion model predicts shape by inclusion of bond angles and most distant arrangement of atoms that leads to minimum repulsion. For the molecules that have no lone pairs around the central atom the bonded-atom unshared -pair arrangement is decided by the table as follows:
In order to determine the shape the steps to be followed are indicated as follows:
- 1. Lewis structure of molecule should be written.
- 2. The type electron arrangement around the central atom should be identified around the central atom. This essentially refers to determination of bond pairs and unshared or lone pairs around central atoms.
- 3. Then bonded-atom unshared -pair arrangement that can maximize the distance of electron pairs about central atom determines the shape.
For molecules that have lone pairs around central atom, lone pairs influence shape, because there are no atoms at the positions occupied by these lone pairs. The key rule that governs the molecular shape, in this case, is the extent of lone –lone pair repulsions are far greater than lone bond pair or bond pair-bond pair repulsions. The table that summarized the molecular shapes possible for various combinations of bonded and lone pairs are given as follows:
(a)
Answer to Problem 2E.16E
The shape for
Explanation of Solution
Total valence electrons are sum of the valence electrons on each atom in
The skeleton structure in
These 15 electron pairs are allotted as lone pairs to satisfy respective octets. Hence, the Lewis structure in
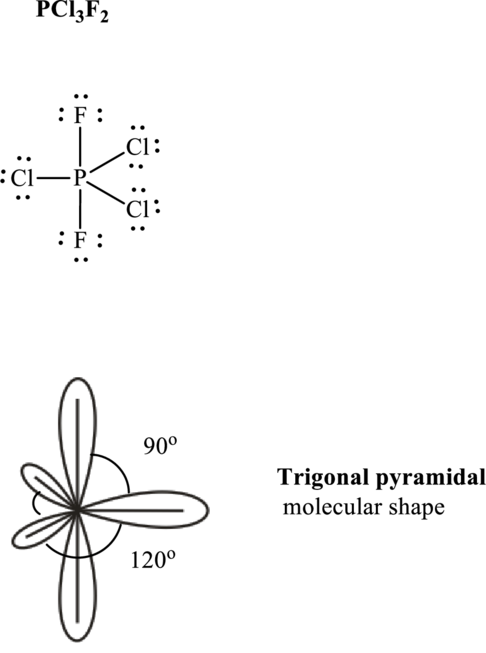
It is evident that
One lone pair is localized on equatorial positions so as to minimize lone pair–bond pair repulsions in accordance with VSPER model. This leads see-saw shape for
If lone pairs are represented by E, central atom with A and each unique atom attached by X and
(b)
Interpretation:
Lewis structure, VSEPR formula, bond angle, and molecular shape for
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 2E.16E
The shape for
Explanation of Solution
Total valence electrons are sum of the valence electrons on atom in
The skeleton structure in
These 12 electron pairs are allotted as lone pairs to satisfy respective octets. Hence, the Lewis structure in
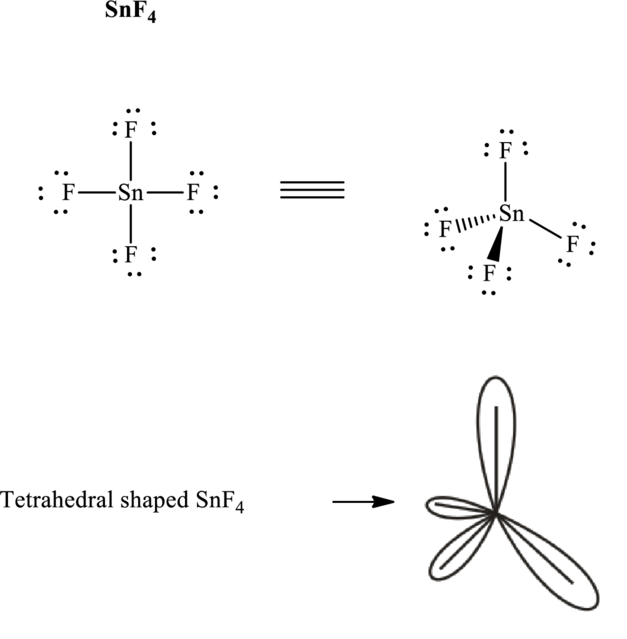
It is evident that
If lone pairs are represented by E, central atom with A and other attached bon pairs by X, then for any tetrahedral species with no one pairs the VSEPR formula is predicted to be
(c)
Interpretation:
Lewis structure, VSEPR formula, bond angle, and molecular shape for
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 2E.16E
The shape for
Explanation of Solution
Total valence electrons are sum of the valence electrons on atom along with two negative charges in
The skeleton structure in
These 18 electron pairs are allotted as lone pairs on each fluorine atom to satisfy respective octets. Hence, the Lewis structure in
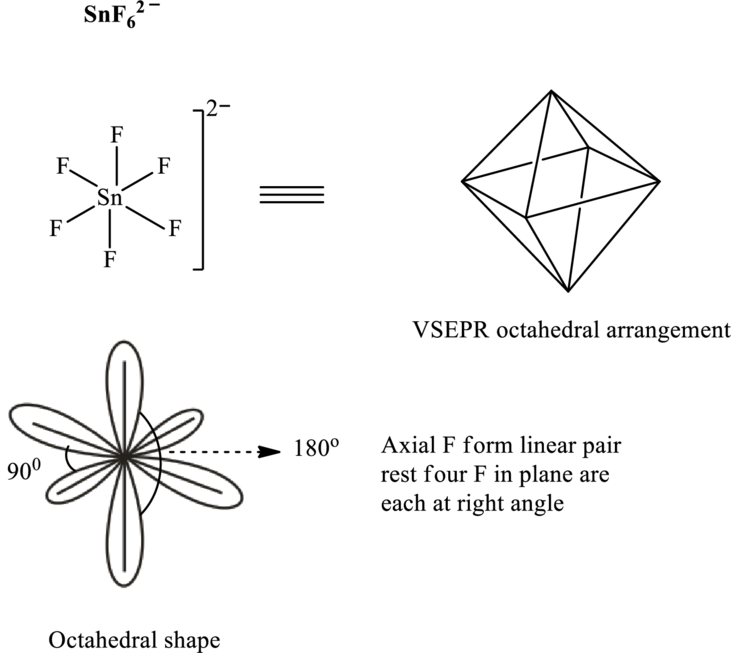
It is evident that in
(d)
Interpretation:
Lewis structure, VSEPR formula, bond angle and molecular shape for
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 2E.16E
The shape for
Explanation of Solution
Total valence electrons are sum of the valence electrons on each fluorine and central iodine in
The skeleton structure in
These 16 electron pairs are allotted as lone pairs of each of the fluorine atoms and one on central iodine to satisfy respective octet. Hence, the Lewis structure
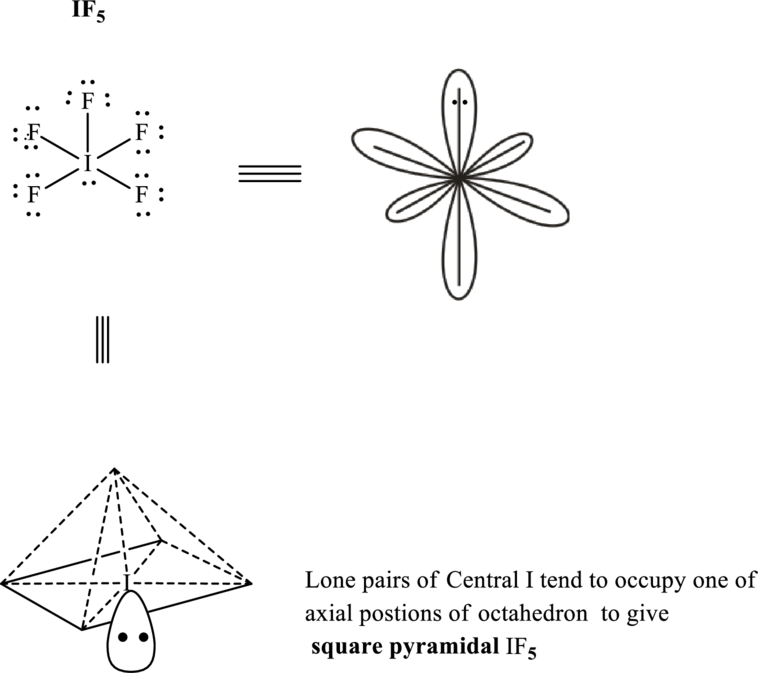
It is evident that in
If lone pairs are represented by E, central atom with A and other attached bond pairs by X, then for any square planar species the VSEPR formula is predicted as
(e)
Interpretation:
Lewis structure, VSEPR formula, bond angle and molecular shape for
Concept Introduction:
Refer to part (a).
(e)
Answer to Problem 2E.16E
The shape for
Explanation of Solution
Total valence electrons are sum of the valence electrons on atom in
Thus, Lewis structure in
These 12 electron pairs are allotted as either lone pairs or multiple bonds with
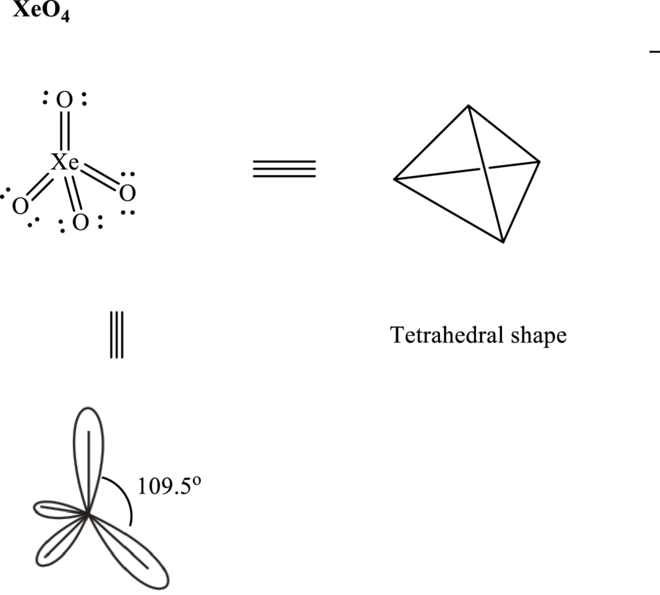
It is evident that in
So
If lone pairs are represented by E, central atom with A and other attached bond pairs by X, then for any tetrahedral species the VSEPR formula is predicted as
Want to see more full solutions like this?
Chapter 2 Solutions
ACHIEVE/CHEMICAL PRINCIPLES ACCESS 1TERM
- Predict die molecular structure and bond angles for each molecule or ion in Exercises 88 and 94. a. POCl3, SO42, XeO4, PO43, ClO4 b. NF3, SO32, PO33, ClO3 c.ClO2, SCl2, PCl2 d. Considering your answers to parts a, b, and c. what conclusions can you draw concerning the structures of species containing the same number of atoms and the same number of valence electrons? (O3), sulfur dioxide, and sulfur trioxide.arrow_forwardIt is possible to write a simple Lewis structure for the SO42- ion, involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs. (a) Draw the two Lewis structures. (b) What geometries are predicted for the two structures? (c) What is the hybridization of sulfur in each case? (d) What are the formal charges of the atoms in the two structures?arrow_forwardIn the following compounds, the C atoms form a single ring.Draw a Lewis structure for each compound, identify cases for which resonance exists, and determine the carbon-carbon bondorder(s): (a) C₃H₄; (b) C₃H₆; (c) C₄H₆; (d) C₄H₄; (e) C₆H₆.arrow_forward
- Consider the reaction BF3 + NH3 -> F3B-NH3 (a) Describe the changes in hybridization of the B and N atoms as a result of this reaction. (b) Describe the shapes of all the reactant molecules with their bond angles. (c) Draw the overall shape of the product molecule and identify the bond angles around B and N atoms. (d) What is the name of the bond between B and N. (e)Describe the bonding orbitals that make the B and F, B and N & N and H bonds in the product molecule.arrow_forwardDraw the Lewis structure with lowest formal charges, and determine the charge of each atom in (a) OCS; (b) NO. (C)CN−; (d) ClO−.arrow_forwardDraw the molecular shapes and predict the bond angles (relative to the ideal angles) of (a) SbF5 and (b) BrF5.arrow_forward
- Help me pleasearrow_forward. Assume that the third-period element phosphorus forms a diatomic molecule, P2, in an analogous way as nitrogen does to form N2. (a) Write the electronic configuration for P2. Use [Ne2] to represent the electron configuration for the first two periods. (b) Calculate its bond order. (c) What are its magnetic properties (diamagnetic or paramagnetic)?arrow_forwardMethyl isocyanate, CH3NCO, was made infamous in 1984when an accidental leakage of this compound from a storagetank in Bhopal, India, resulted in the deaths of about3800 people and severe and lasting injury to many thousandsmore. (a) Draw a Lewis structure for methyl isocyanate.(b) Draw a ball-and-stick model of the structure,including estimates of all the bond angles in the compound.(c) Predict all the bond distances in the molecule.(d) Do you predict that the molecule will have a dipolemoment? Explain.arrow_forward
- Determine whether each molecule is polar or nonpolar: (a) H 2O; (b) CO 2.arrow_forward3. Provide the correct Lewis representation of (a) sodium chloride formation and (b) aluminum fluoride. 4. Provide the (a) Lewis Structure, (b) hybridization, and (c) molecular geometry of XeF2, H3O*, and SnBr4.arrow_forwardConsider the following molecular shapes:a) Match each shape with one of the following species: XeF₃,SbBr₃, GaCl₃. (b) Which, if any, is polar? (c) Which has the most valence electrons around the cental atom?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


