
(a)
Interpretation:
The
Concept Introduction:
The fundamental principles that are followed to write an electronic configuration include three rules as follows:
Electron in a
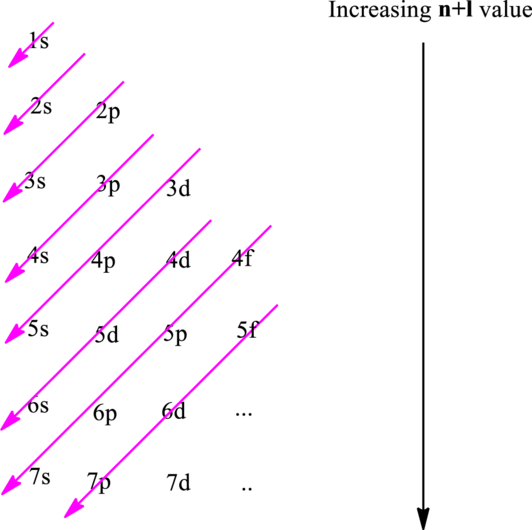
Hund’s rule suggests electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
Pauli Exclusion Principle states two electrons within the same orbital cannot possess same set for four possible quantum numbers. Quantum-mechanical equivalent for this state means that two electrons are not allowed to have the same space simultaneously. Since one electron may certain discrete same set of n, l and m values, however, the fourth spin quantum number should be different. This provides a unique address to every electron. Since the possible magnitudes of spin can be either +1/2 or -1/2 thus at maximum two electrons can occupy any given orbital.
The convention followed to remove or add electrons is electrons of largest principal quantum number are lost first. In case of subshells of the same
(b)
Interpretation:
The
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
ACHIEVE/CHEMICAL PRINCIPLES ACCESS 1TERM
- Mixing SbCl3 and GaCl3 in a 1:1 molar ratio using liquid sulfur dioxide as a solvent gives a solidionic compound with the empirical formula GaSbCl6. A controversy arose over whether this compoundis [SbCl2]+[GaCl4]− or [GaCl2]+[SbCl4]−.(a) Predict the molecular structure of the two anions from the two choices using VSEPR theory.(b) It is learned that the cation in the compound has a bent structure. Based on this fact, whichformulation is the correct one?arrow_forwardDefine the following phenomena with reference to main group compounds: (d) multicenter electron deficient bondingarrow_forwardWrite the electron configuration for each of the following ions and determine which one possess noble-gas configurations: (a) Sr2+; (b) Ti2+; (c) Se2-; (d) Ni2+; (e) Br-; (f) Mn3+.arrow_forward
- Which member of each pair has more covalentcharacter inits bonds: (a) LiCl or KCl; (b) AlCl₃ or PCl₃; (c) NCl₃ or AsCl₃?arrow_forwardUse the data given below to calculate the bond energies (Do) (in eV) of1H35Cl and 1H81Br molecules. Which bond is stronger? HCl Ve (cm^-1): 2991, De(cm^-1): 53194 HBr Ve(cm^-1):2649 , De(cm^-1): 34570arrow_forwardWrite the electron configurations for the following ions, anddetermine which have noble-gas configurations: (a) Co2+ ,(b) Sn2+ , (c) Zr4+ , (d) Ag+, (e) S2- .arrow_forward
- Determine the average Cl-F bond energy, in units of kJ mol¯, in CIF5, using t following data: • Cl2(g) + 5 F2(g) → 2 CIF5(g) A,H° = -477 kJ mol-1 159 kJ mol-1 • bond energy F-F: • bond energy Cl-Cl: 243 kJ mol-1arrow_forward(c) Draw the orbital diagrams and Lewis symbols to depict the formation of Na* and CI ions from the atoms. Give the formula of the compound formed. (d) The predicted bond length for HF is 109 pm (the sum of the covalent radii of H, 37 pm and F. 72 pm), however the actual bond length for HF is shorter (92 pm). It was observed that the difference between predicted and actual bond lengths becomes smalleor going down the halogen group from HF to HI Describe these observationsarrow_forwardAluminum chloride, AlCl₃, is used in a variety of organic reactions as a Lewis acid. Although it adopts a more complex geometry in the solid state, consider it as a single molecule (i.e., in the gas phase). Given the electron configuration of Al is [Ne]3s²3p¹, how many valence electrons does Al have?arrow_forward
- Write the electron configurations for: Calcium ion in CaCl₂:arrow_forwardRank the following in increasing order of nonmetallic character by numbering them in order from 1 through 6, with 1 being the least nonmetallic and 6 being the most nonmetallic. Se Cr K Br Са Asarrow_forwardConsider the following electron configuration:(σ3s)2(σ3s*)2(σ3p)2(π3p)4(π3p*)4Give four species that, in theory, would have this electron configuration.arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

