
Concept explainers
(a)
Interpretation:
Valence shell electronic configuration of
Concept Introduction:
Molecular orbital diagram is a linear combination of atomic orbitals of similar energy and similar symmetry. It is formed by the proper overlap of the atomic orbitals.
There are 3 types of molecular orbitals as follows:
1. Bonding molecular orbital: They are formed by the constructive interference of atomic orbitals and electrons in it stabilize the molecule and are of lesser in energy.
2. Antibonding molecular orbital: This type of orbitals increases the energy of molecule and destabilizes it and weakens the bond between the atoms.
3. Non-bonding molecular orbital: These types of orbitals have energy similar to atomic orbitals that is addition or removal of electron does not change the energy of molecule.
The order of energy in molecular orbital follows two rules as follows:
1. For
2. For atomic number more than 14 order of energy is,
(a)
Explanation of Solution
For
The symbol for nitrogen is
One positive charge is added up in the total valence count.
Thus total valence electrons is sum of the valence electrons for each atom in
Hence, 9 electrons are to be arranged in each molecular orbital to obtain an electronic configuration. Since number of electrons in
Hence, the electronic configuration of
For
The symbol for nitrogen is
Two positive charges are subtracted from the total valence count.
Thus total valence electrons are sum of the valence electrons for each atom in
Hence, 8 electrons are to be arranged in each molecular orbital to obtain an electronic configuration. Since number of electrons in
Hence, the electronic configuration of
For
The symbol for nitrogen is
Two negative charges are added up in total valence count.
Thus total valence electrons are sum of the valence electrons for each atom in
Hence, 12 electrons are to be arranged in each molecular orbital to obtain an electronic configuration. Since number of electrons in
Hence, the electronic configuration of
(b)
Interpretation:
The bond order of
Concept Introduction:
Bond order
(b)
Explanation of Solution
For
The electronic configuration of
Substitute 7 for number of electrons in bonding orbitals and 2 for number of electrons in antibonding orbitals in equation (1) to calculate bond order.
Hence, the bond order of the molecule
For
The electronic configuration of
Substitute 6 for number of electrons in bonding orbitals and 2 for number of electrons in antibonding orbitals in equation (1) to calculate bond order.
Hence, the bond order of the molecule
For
The electronic configuration of
Substitute 8 for number of electrons in bonding orbitals and 4 for number of electrons in antibonding orbitals in equation (1) to calculate bond order.
Hence, the bond order of the molecule
(c)
Interpretation:
Molecular orbital diagram of
Concept Introduction:
Refer to (a)
(c)
Explanation of Solution
For
The electronic configuration of
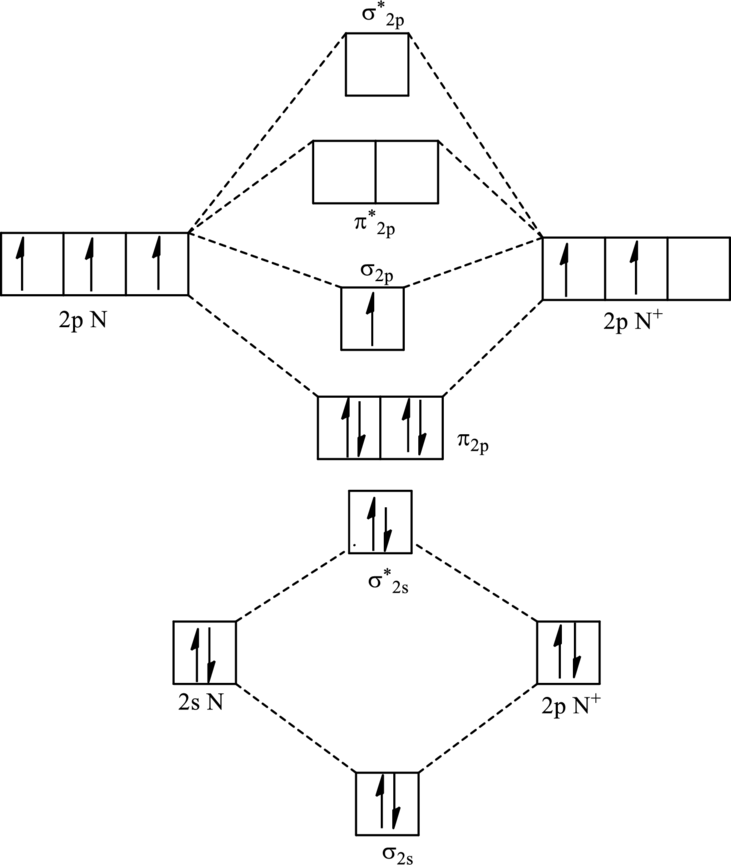
Since
For
The electronic configuration of
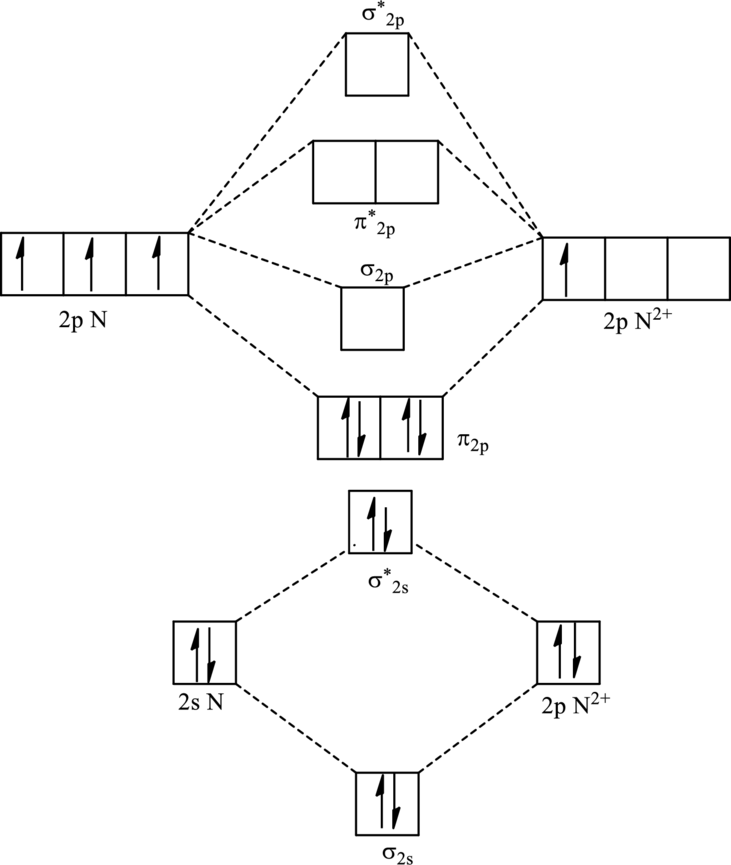
Since
For
The electronic configuration of
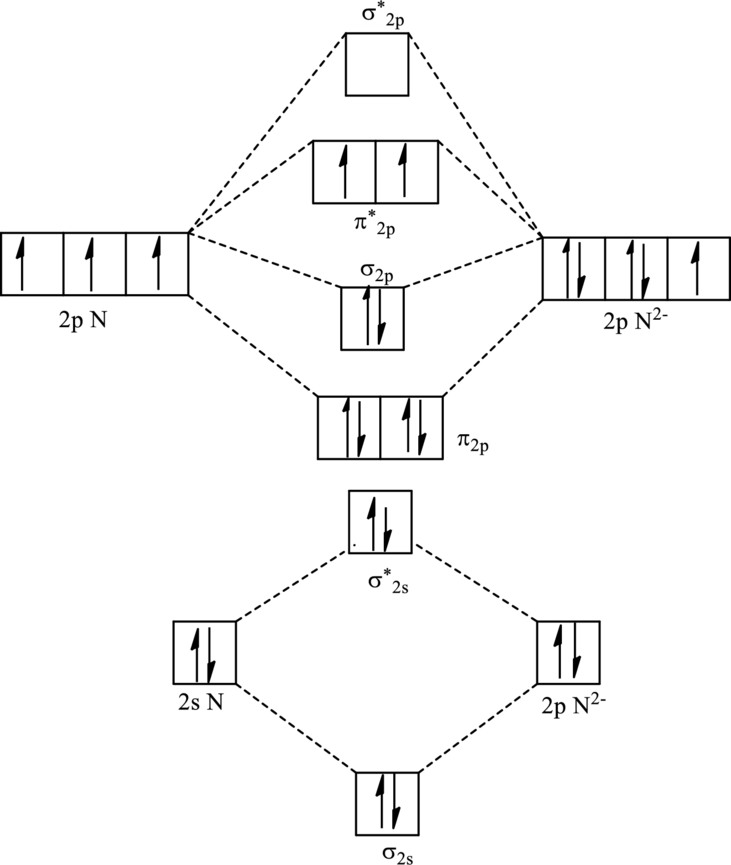
Since
(d)
Interpretation:
Character of highest energy orbital of
Concept Introduction:
Refer to (a)
(d)
Explanation of Solution
For
The electronic configuration of
For
The electronic configuration of
For
The electronic configuration of
Want to see more full solutions like this?
Chapter 2 Solutions
ACHIEVE/CHEMICAL PRINCIPLES ACCESS 1TERM