
Interpretation:
To explain the difference in the observed pK values for P and Q.
Concept introduction:
Acid base reaction or Proton transfer reaction. It is the equilibrium reaction. An acid donates a proton to a based and thus forming conjugate acid and base. The reaction is always involves two acid and bases. It is equilibrium reaction so the position of the equilibrium depends on the strength of the acid and bases. The stronger is the acid that donates proton and the stronger base accept the proton. Then these two forms the conjugate acid and base as shown below in the Fig.
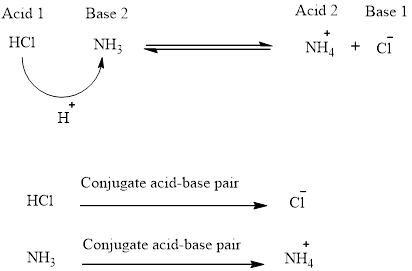
From the Bronsted-Lowry, an acid is a proton donor and from the Lewis electron pairs acceptor. Thus an acid strength is defined as the tendency to donate proton. The decision of an acid is stronger is based on the compound donating proton. If the compound readily donates the proton then it is strong acid. The strong acid is determined using the acidity constant, Ka. So larger the Ka value stronger the acid. Moreover for exponent value it is conveniently measured by pKa values.
pKa = -log Ka
So, smaller the pKa, stronger is the acid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Organic Chemistry
- If the transverse relaxation time, T2, is 50ms, after what time will theenvelope of the free induction decay decrease to half its initial amplitude?arrow_forwardthank you so much, can u answer f, g and 6 in a separate answer?arrow_forwardCalculate the separation factor (or selectivity factor), between two compounds, 1 and 2, whose retention volumes are 6ml and 7ml, respectively. The dead volume of the column used is 1ml.arrow_forward
- Q1-) Give information about the reasons why instrumental analysis techniques are preferred over classical analysis techniques.arrow_forwardWhat is the difference in population of 13C spins between the upper and lower states in a magnetic field at 2T and 300K?arrow_forwardWhat is the name of the standard that defines zero on the chemical shift axis? (The standard must be a molecule with protons in only one type of environment.) -Tetramethylsilane -Propanoic acid -Ethanol -Butanearrow_forward
- 5- The vibrations in infrared spectroscopy might be active or inactive?arrow_forwardA)A molecule which possesses an inversion center is said to be centrosymmetric. We have seen that IR and Raman spectroscopy can be used to distinguish between alternative structures of molecules. If one is considering two possible structures of a molecule, one of which is centrosymmetric and one of which is not (for example, a molecule with formula ML4 could be square-planar or tetrahedral), the following rule is useful. In a centrosymmetric molecule, a given vibrational mode cannot be active in BOTH the IR and Raman spectrum, while in a non-centrosymmetric molecule, there is no such exclusion. Look at the character tables for a number of point groups and justify this rule.arrow_forwardIf a population has 5261 members and you select a sample of 45 using systematic sampeling, what is the value of m? Iarrow_forward
- 2. I don't exactly understand what this question is trying to do or ask or how to even solve it. What does the Gcalc, Gtable, Qcalc and Qtable even mean in this context?arrow_forwardExpress the π electron binding energy of benzene in terms of the Huckel parameters α and β.arrow_forward-1 56 The first three Stokes lines in the rotational Raman spectrum of ¹0₂ are separated by 14.4 cm¹, 25.8 cm¹ and 37.4 cm from the exciting radiation. Using the rigid rotor approximation obtain a value for ro.arrow_forward
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning


