
Organic Chemistry
4th Edition
ISBN: 9780073402772
Author: Janice G. Smith
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2.47P
Which compounds can be deprotonated by
(a).
(b).
(c). 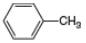
(d).
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
4. Consider the following bases: Which of the following bases is strong enough to deprotonate
CH4? Explain based on the direction of equilibrium:
a
b
H
H
·3.
Consider the following acidic equilibrium: H₂CO₃(aq) + H₂O(l) ⇌ HCO₃⁻(aq) + H₃O⁺(aq). If you add NaHCO₃ to this solution, which of the following will occur?
A) The reaction quotient will decrease.
B) The reaction will shift in the reverse direction.
C) The equilibrium constant will increase.
D) No changes to the equilibrium positions will take place.
Swimming pool disinfectants produce hypochlorous acid upon dissolution. The weak acid ionizes as follows:
HClO (aq) ⇄ H+ (aq) + ClO‒ (aq) Ka=3.0 x 10 ‒8
As strong oxidizing agents, both acid and its conjugate base kill bacteria. However, too high [HClO] is irritating to swimmers’ eyes and too high [ClO‒] will cause the ions to decompose in sunlight. The recommended pH to circumvent both problems is 7.8. Determine the ratio of the weak acid and its conjugate base at this pH.
Chapter 2 Solutions
Organic Chemistry
Ch. 2 - a. Which compounds are Bronsted-Lowry acids:...Ch. 2 - a. Draw the conjugate acid of each base:...Ch. 2 - Label the acid and base, and the conjugate acid...Ch. 2 - Draw the products of each proton transfer...Ch. 2 - Draw the products formed from the acid-base...Ch. 2 - Which compound in each pair is the stronger acid?...Ch. 2 - Use a calculator when necessary to answer the...Ch. 2 - Rank the conjugate bases of each of group of acids...Ch. 2 - Problem-2.10 Considers two acids: (formic acid,)...Ch. 2 - Estimate the pKa of each of the indicated bonds.
Ch. 2 - Draw the products of each reaction and determine...Ch. 2 - Prob. 2.12PCh. 2 - Without reference to a pKa table, decide which...Ch. 2 - which compound in each pair of isomers is the...Ch. 2 - Prob. 2.15PCh. 2 - Which hydrogen in pseudoephedrine, the nasal...Ch. 2 - whichcompound in each pair is the stronger acid? a...Ch. 2 - Glycolic acid, HOCH2CO2H, is the simplest member...Ch. 2 - Explain the apparent paradox. HBr is a stronger...Ch. 2 - The CH bond in acetone, (CH3)2C=O, has a pKa of...Ch. 2 - Acetonitrile (CH3CN) has a pKa of 25, making it...Ch. 2 - For each pair of compounds: [1] Which indicated H...Ch. 2 - Rank the compounds in each group in order of...Ch. 2 - Which proton in each of the following drugs is...Ch. 2 - Prob. 2.25PCh. 2 - Problem 2.29
Compounds like amphetamine that...Ch. 2 - Problem 2.30 Which species are Lewis bases?
a. b....Ch. 2 - Which species are Lewis acids?
a. b. c. d.
Ch. 2 - For each reaction, label the Lewis acid and base....Ch. 2 - Prob. 2.30PCh. 2 - Prob. 2.31PCh. 2 - Prob. 2.32PCh. 2 - 2.36 Propranolol is an antihypertensive agent—that...Ch. 2 - 2.37 Amphetamine is a powerful stimulant of the...Ch. 2 - Prob. 2.35PCh. 2 - Prob. 2.36PCh. 2 - a Draw the conjugate acid of ethylene, CH2 = CH2....Ch. 2 - 2.40 Draw the products formed from the acid-base...Ch. 2 - Draw the products formed from the acid-base...Ch. 2 - Prob. 2.40PCh. 2 - Draw the product of acid-base reaction. a. c. b....Ch. 2 - Prob. 2.42PCh. 2 - Prob. 2.43PCh. 2 - What is Ka for each compound? Use a calculator...Ch. 2 - What is the pKa for each compound? a. b. c.Ch. 2 - Which of the following bases are strong enough to...Ch. 2 - Which compounds can be deprotonated by OH, so that...Ch. 2 - Draw the product of each reaction. Use the pKa...Ch. 2 - Rank the following compounds in order of...Ch. 2 - Rank the following ions in order of increasing...Ch. 2 - Prob. 2.51PCh. 2 - Prob. 2.52PCh. 2 - The pKa of three CH bonds is given below. a. For...Ch. 2 - a. What is the conjugate acid of A? b. What is the...Ch. 2 - 2.56 Draw the structure of a constitutional isomer...Ch. 2 - 2.57 Many drugs are Bronsted-Lowry acids or...Ch. 2 - Dimethyl ether (CH3OCH3) and ethanol (CH3CH2OH)...Ch. 2 - Prob. 2.58PCh. 2 - Ethyl butanoate, CH3CH2CH2CO2CH2CH3, is one of the...Ch. 2 - Prob. 2.60PCh. 2 - 2.61 Label the three most acidic hydrogen atoms in...Ch. 2 - Prob. 2.62PCh. 2 - 2.64 Classify each species as a Lewis acid, a...Ch. 2 - Prob. 2.64PCh. 2 - Draw the products of each Lewis acid-base...Ch. 2 - Prob. 2.66PCh. 2 - Prob. 2.67PCh. 2 - 2.70 Hydroxide can react as a Brønsted-Lowry base...Ch. 2 - 2.71 Answer the following questions about esmolol,...Ch. 2 - Prob. 2.70PCh. 2 - 2.72 DBU, is a base we will encounter in...Ch. 2 - 2.73 Molecules like acetamide can be protonated...Ch. 2 - Two pKa values are reported for malonic acid, a...Ch. 2 - Prob. 2.74PCh. 2 - 2.76 Write a stepwise reaction sequence using...Ch. 2 - Prob. 2.76PCh. 2 - 2.78 Which compound, M or N, is the stronger acid?...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Most naturally occurring acids are weak acids. Lactic acid is one example. CH3CH(OH)CO2H(s)+H2O(l)H3O+(aq)+CH3CH(OH)CO2(aq) If you place some lactic acid in water, it will ionize to a small extent, and an equilibrium will be established. Suggest some experiments to prow that this is a weak acid and that the establishment of equilibrium is a reversible process.arrow_forwardYou are on one of Saturn's many moons and discover a substance, Y2O that undergoes autoionization just like water on Earth. Y2O + Y2O ⇌ Y3O+ + OY– Through a series of experiments, you determine the equilibrium constant (Keq) for this autoionization reaction at various temperatures. The value of Keq at 28.7ºC is 7.6 x 10-14. What is the pY of the pure substance at this temperature? Report your answer to the hundreths placearrow_forwardPotassium hydroxide (KOH) is a strong base. Dissociation of potassium hydroxide follows the reaction equation given. KOH(s) --H20--> K+(aq) OH-(aq) a) Write down the reaction equation for a neutralization reaction where potassium hydroxide (KOH) reacts with sulphuric acid (H2SO4) producing potassium sulphate (K2SO4) and water (H2O). Balance if needed! b) Calculate pH for the solution where 0,2 mol of KOH is dissolved into water so that the total volume of the solution is 0,5 L.arrow_forward
- Predict whether aqueous solutions of the following substances are acidic, basic, or neutral and write hydrolysis equations for the acidic and basic solutions. (a) CsBr; (b) Al(NO3)3; (c) KCN; (d) CH3NH3Clarrow_forward1. Nitrogen in an industrial wastewater is primarily in the form of ammonia (NH3) and ammonium (NH4+) ions. The acid/base equilibrium reaction for ammonia and ammonium is given as: NH4+ → H+ + NH3 Ką = 10-9.26 The total nitrogen concentration in wastewater is given as 2 x 10-3 moles/L (or 28 mg N/L), a. Calculate the concentration of ammonia (NH3) and ammonium (NH4*) ions at pH 10. NH3: moles/L NH4*: moles/L Check b. In ammonia stripping, nitrogen is removed from wastewater by volatilization of NH3. Would ammonia stripping be more effective above or below pH 9? Briefly explain your reasoning.arrow_forwardWrite the equilibrium constant expression, K, for the following reaction taking place in dilute aqueous solution.ClO- (aq) + H2O (l)HClO (aq) + OH- (aq)arrow_forward
- You found a bottle of aqueous solution in the laboratory cabinet. Unfortunately, the label has been eroded and you could not recognize it. To the best of your recollection, it may be one of the following solutions: HCl CH3COOH CH3CH2COOH A mixture of HF and NaF (both of substantial amount) A mixture of H3PO4 and NaH2PO4 (both of substantial amount) NH4Cl NaHCO3 In order to identify the solution, you conduct the following experiments: Using a pH meter, you determine the pH of the solution to be 3.00. You dilute 20 mL of the solution with water to a total volume of 200 mL and measure the pH again, this time it reads 3.50. You take some volume of the solution, add phenolphthalein, and titrate it with NaOH solution until the mixture turns pink. You record the volume of the required titrant as Vt and the pH meter reads 9.05. In a separate flask, you take the same volume of the unknown solution as in step iii and titrate it with the same NaOH…arrow_forwardYou found a bottle of aqueous solution in the laboratory cabinet. Unfortunately, the label has been eroded and you could not recognize it. To the best of your recollection, it may be one of the following solutions: HCl CH3COOH CH3CH2COOH A mixture of HF and NaF (both of substantial amount) A mixture of H3PO4 and NaH2PO4 (both of substantial amount) NH4Cl NaHCO3 In order to identify the solution, you conduct the following experiments: Using a pH meter, you determine the pH of the solution to be 3.00. You dilute 20 mL of the solution with water to a total volume of 200 mL and measure the pH again, this time it reads 3.50. You take some volume of the solution, add phenolphthalein, and titrate it with NaOH solution until the mixture turns pink. You record the volume of the required titrant as Vt and the pH meter reads 9.05. In a separate flask, you take the same volume of the unknown solution as in step iii and titrate it with the same NaOH…arrow_forwardYou found a bottle of aqueous solution in the laboratory cabinet. Unfortunately, the label has been eroded and you could not recognize it. To the best of your recollection, it may be one of the following solutions: HCl CH3COOH CH3CH2COOH A mixture of HF and NaF (both of substantial amount) A mixture of H3PO4 and NaH2PO4 (both of substantial amount) NH4Cl NaHCO3 In order to identify the solution, you conduct the following experiments: i.Using a pH meter, you determine the pH of the solution to be 3.00. ii. You dilute 20 mL of the solution with water to a total volume of 200 mL and measure the pH again, this time it reads 3.50. iii. You take some volume of the solution, add phenolphthalein, and titrate it with NaOH solution until the mixture turns pink. You record the volume of the required titrant as Vt and the pH meter reads 9.05. iv. In a separate flask, you take the same volume of the unknown solution as in step iii and titrate it with the same NaOH solution, but this time…arrow_forward
- You found a bottle of aqueous solution in the laboratory cabinet. Unfortunately, the label has been eroded and you could not recognize it. To the best of your recollection, it may be one of the following solutions: HCl CH3COOH CH3CH2COOH A mixture of HF and NaF (both of substantial amount) A mixture of H3PO4 and NaH2PO4 (both of substantial amount) NH4Cl NaHCO3 In order to identify the solution, you conduct the following experiments: Using a pH meter, you determine the pH of the solution to be 3.00. You dilute 20 mL of the solution with water to a total volume of 200 mL and measure the pH again, this time it reads 3.50. You take some volume of the solution, add phenolphthalein, and titrate it with NaOH solution until the mixture turns pink. You record the volume of the required titrant as Vt and the pH meter reads 9.05. In a separate flask, you take the same volume of the unknown solution as in step iii and titrate it with the same NaOH…arrow_forwardYou found a bottle of aqueous solution in the laboratory cabinet. Unfortunately, the label has been eroded and you could not recognize it. To the best of your recollection, it may be one of the following solutions: HCl CH3COOH CH3CH2COOH A mixture of HF and NaF (both of substantial amount) A mixture of H3PO4 and NaH2PO4 (both of substantial amount) NH4Cl NaHCO3 In order to identify the solution, you conduct the following experiments: 1. Using a pH meter, you determine the pH of the solution be 3.00. 2. You dilute 20 mL of the solution with water to a total volume of 200 mL and measure the pH again, this time it reads 3.50. 3. You take some volume of the solution, add phenolphthalein, and titrate it with NaOH solution until the mixture turns pink. You record the volume of the required titrant as Vt and the pH meter reads 9.05. ti =0.0 and tf=2.0. 4. In a separate flask, you take the same volume of the unknown solution as in step iii and titrate it with the same NaOH…arrow_forwardOrganic chemistry:Hydrochloric acid is about as strong a mineral acid as sulfuric acid .Why would HCL not be a satisfactory catalyst in the synthesis of an aspirin?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
ENVIRONMENTAL POLLUTION; Author: 7activestudio;https://www.youtube.com/watch?v=oxtMFmDTv3Q;License: Standard YouTube License, CC-BY