
Concept explainers
Show by a series of equations how could synthesize each of the following compounds from the indicated starting material and any necessary organic or inorganic reagents:
(a)
(b)
(c)
(d)
(e)
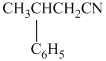
(f) 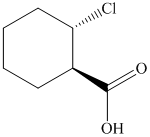 from
from 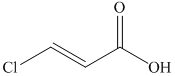
(g)
(h)
(i)
Interpretation:
The way in which each of the given compounds can be synthesized from the indicated starting material and any necessary organic or inorganic reagents is to be shown by using a series of equations.
Concept Introduction:
Hydroboration reaction is a two-step reaction that involves conversion of an alkene into alcohol. This type of reaction follows anti-Markovnikov's rule.In acid-catalyzed dehydration, a saturated compound is converted to an unsaturated compound with the removal of a water molecule in the presence of an acid catalyst.
Grignard reagent is prepared by the reaction of alkyl or aryl bromide with magnesium metal in the presence of ether.
Thionyl chloride
Lithium aluminium hydride is a strong reducing agent. The reduction of carboxylic acid by
Oxidation reaction involves increase in the
Answer to Problem 18P
Solution:
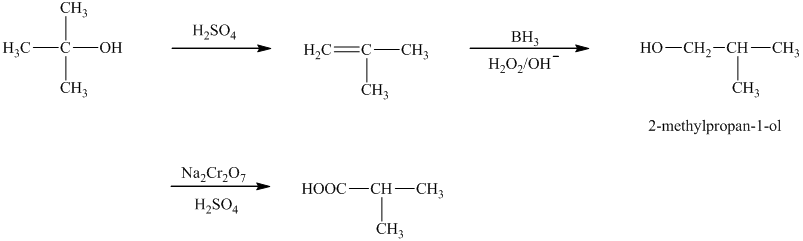
a)The reaction that shows the preparation of
b)The reaction that shows the preparation of
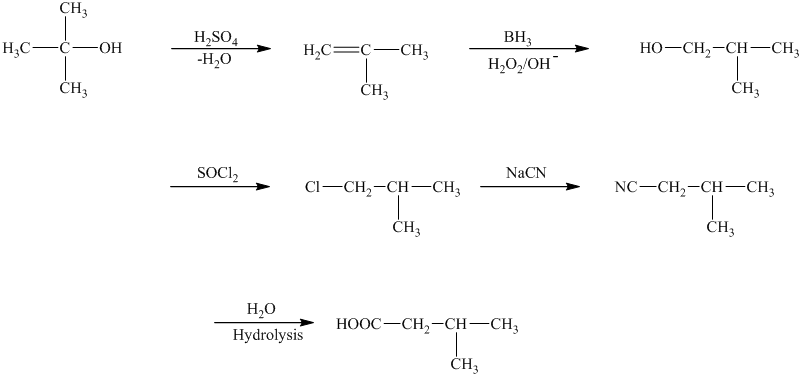
c)The reaction that shows the preparation of
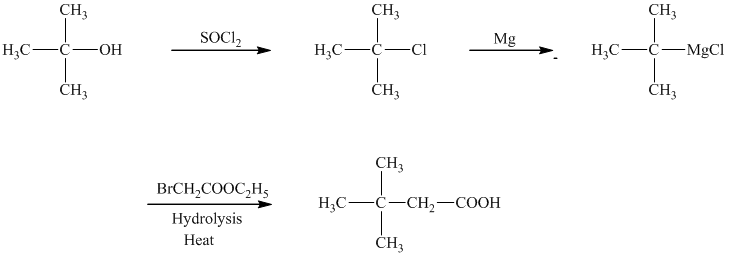
d)The reaction that shows the preparation of
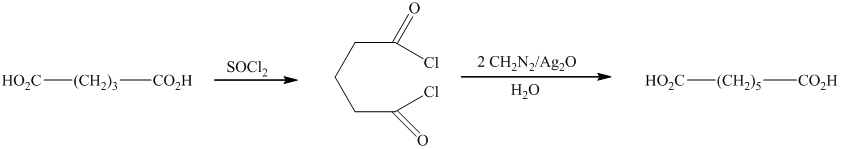
e) The reaction that shows the preparation of
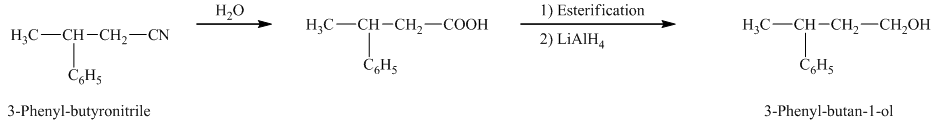
f) The reaction that shows the preparation of
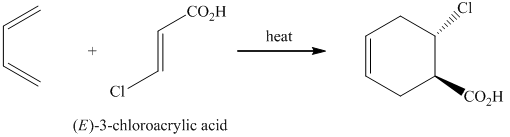
g) The reaction that shows the preparation of
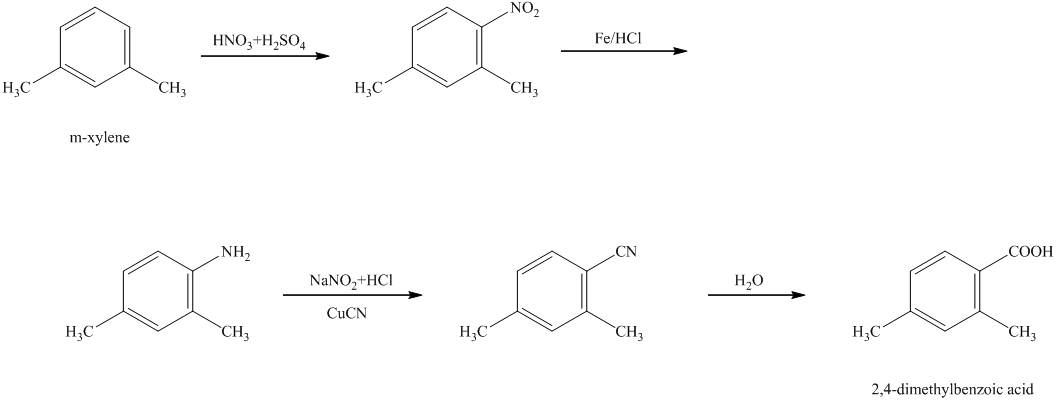
h) The reaction that shows the preparation of
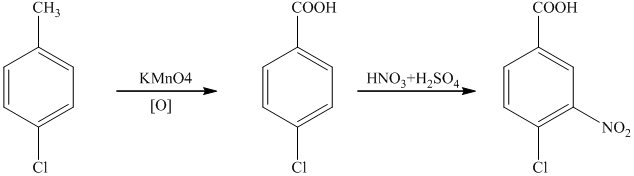
i) The reaction that shows the preparation of
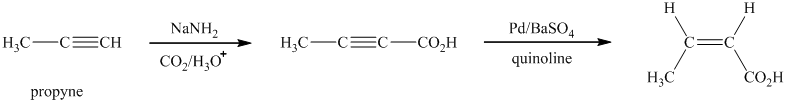
Explanation of Solution
a)
In the synthesis of
Thus, the reaction that shows the preparation of

b)
The reaction that shows the preparation of
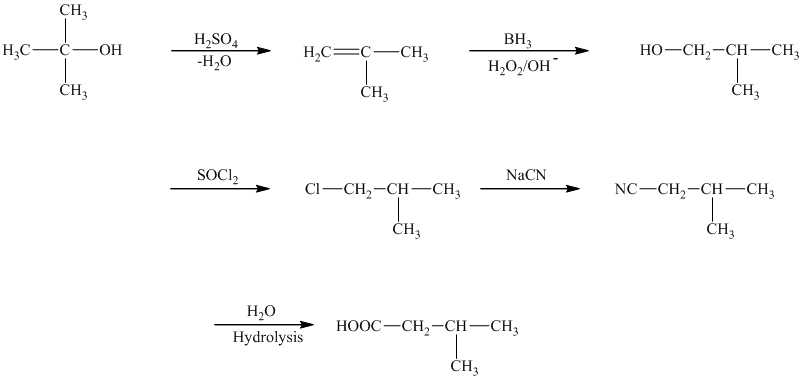
The first step of the required synthesis is the reaction of acid with the given alcohol. This step is the dehydration of alcohol. In the next step, hydroboration of alkene is done, which is then followed by hydrolysis. This results in the formation of
Thus, the required acid is synthesized.
c)
In the required synthesis, the first step is the reaction of the given alcohol with
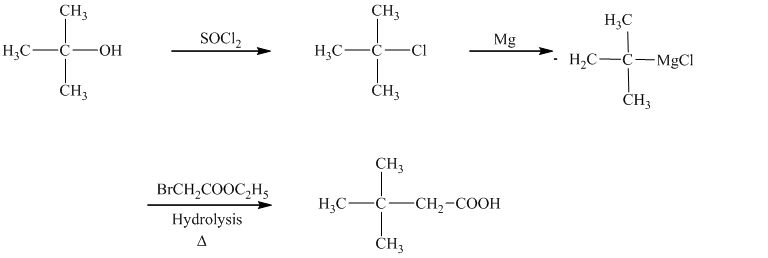
d)
In this synthesis, the first step is the reaction of the given carboxylic acid with thionyl chloride to form acyl chloride. In the next step, acyl chloride reacts with diazomethane to form diazoketones. In the last step of the synthesis reaction, the diazoketones give the final product in the presence of metal catalyst and water. The given reaction is an example of Arndt-Eistert synthesis.
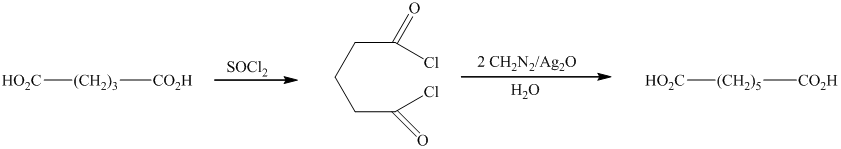
Therefore, the required product is synthesized.
e) 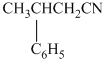
In the required synthesis, the first step is the hydrolysis of
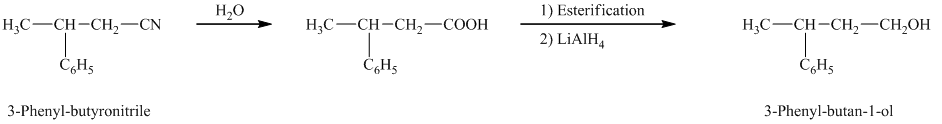
Therefore, the required product is synthesized.
f) 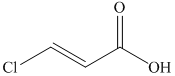
The reaction of
Therefore, the required product is synthesized.
g)
In the required synthesis, the first step is the nitration of
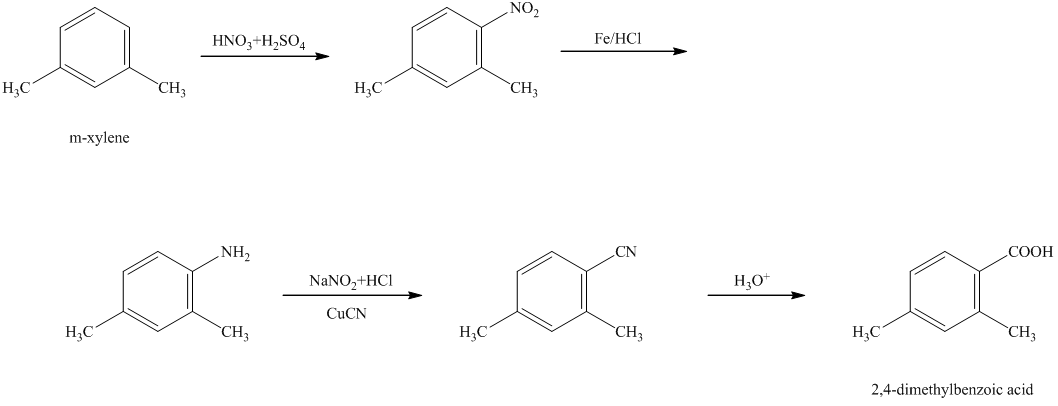
Thus, the required product was synthesized.
h)
In the synthesis of
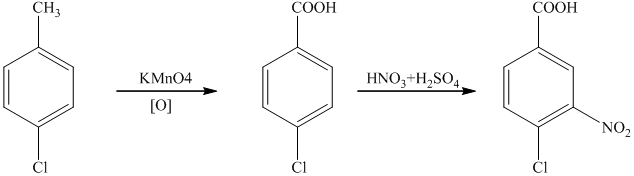
Therefore, the given compound was synthesized.
i)
Sodium amide is a strong base and acts as s strong nucleophile.The reaction that shows the preparation of
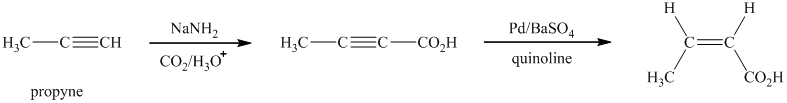
In the above synthesis, the first step is the reaction of
Want to see more full solutions like this?
Chapter 19 Solutions
Organic Chemistry - Standalone book
- Treatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.arrow_forwardHow would you prepare the following compounds from benzene, using a diazonium replacement reaction in your scheme? (a) p-Bromobenzoic acid (b) m-Bromobenzoic acid (c) m-Bromochlorobenzene (d) p-Methylbenzoic acid (e) 1, 2, 4-Tribromobenzenearrow_forwardA chemist in need of 2,2-dimethylpentanoic acid decided to synthesize some by reaction of 2-chloro-2-methylpentane with NaCN, followed by hydrolysis of the product. After the reaction sequence was carried out, however, none of the desired product could be found. What do you suppose went wrong?arrow_forward
- One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forward(a) Arrange the following compounds in an increasing order of their indicated property :(i) Benzoic acid, 4-Nitrobenzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)(ii) CH3CH2CH (Br) COOH, CH3CH (Br) CH2COOH,(CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)(b) How would you bring about the following conversions :(i) Propanone to Propene (ii) Benzoic acid to Benzaldehyde(iii) Bromobenzene to 1-phenylethanolarrow_forward
- Propose the mechanisms for each step of the synthesis below.arrow_forwardHow do you synthesize 3-oxocyclohexanecarboxylic acid from 2-cyclohexenone?give earrow_forward5. 2. When 2,2-dimethyl-1-propanol is heated with acid, it is slowly converted into an 85:15 mixture of 2 alkenes of molecular formula C2H10.a) Give the mechanisms by which they are formed. b) Give the structures of these alkenes. c) Identify with reason which would be the major productarrow_forward
- Diethyl carbonate serves as a less-toxic alternative to phosgene for making Lexan. Propose a mechanism for the transesterification of diethyl carbonate with bisphenol A, catalyzed by a trace of sodium ethoxide. What small molecule is given off in this condensation?arrow_forwardShow how you would synthesize the following compounds, starting with benzene or toluene and any necessary acyclic reagents. Assume para is the major product (and separable from ortho) in ortho, para mixtures.(a) p-aminobenzoic acid (b) 2-methyl-1-phenylbutan-2-ol (c) 5-chloro-2-methylanilinearrow_forwardPredict the products of reactions of ketones and aldehydes with the followingtypes of compounds, and give mechanisms where appropriate: (a) hydridereducing agents; (b) Clemmensen and Wolff-Kishner reagents; (c) Grignard andorganolithium reagents; (d) phosphorus ylides; (e) water; (f) hydrogen cyanide;(g) ammonia and primary amines; (h) hydroxylamine and hydrazine derivatives;(i) alcohols; and (j) oxidizing agents.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

