
Practice Problem CONCEPTUALIZE
CONCEPTUALIZE
Consider the gas-phase reaction of
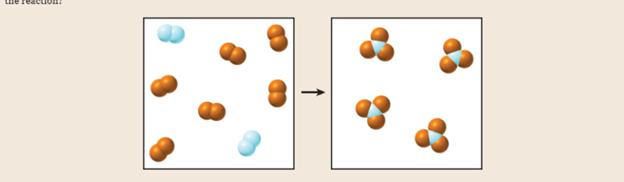
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Chemistry
- For which reaction will ΔS be positive? Group of answer choices N2O4(g) ⟶ 2 NO2(g) 2 H2(g) + O2(g) ⟶2 H2O(g) 3 NO(g) ⟶NO2(g) + N2O(g) 2 SO2(g) + O2(g) ⟶2 SO3(g)arrow_forwardThe ΔG°rxn for this reaction is -107.8 kJ/mol. Given the values for ΔGf°; what is the ΔGf° for A2B(l)? 2AC(g) + B2(g) → A2B(l) + BC2(g) Substance ΔGf° (kJ/mol) AC(g) -174.3 B2(g) 173.3 BC2(g) 169.9arrow_forwardWhich of the following processes have a ΔS that is positive? Select all that apply Group of answer choices H2O (l) → H2O (g) H2O (l) → H2O (s) Al (s) → Al (l) N2 (g) + 3 H2 (g) → 2 NH3 (g) CO2 (s) → CO2 (g)arrow_forward
- What is the sign of Δ S for the reaction below as written? N2O4(g) ---------> N2(g) + 2 O2(g) Group of answer choices positive negative zeroarrow_forward1. Hydrogen gas is dangerous because it explodes spontaneously and explosively. In the hydrogen gas explosion reaction: 2 H2 (g) + O2 (g) → 2 H2O (liq), what are the signs of A,H, A„S, and A,G for this chemical process? a) A,H 0, A,G 0, A,G > 0 A,H> 0, A„S > 0, A,G 0, A,S 0 c) d) e)arrow_forwardWhat is the sign of Δ S for the reaction below as written? CaCO3(s) ---------> CaO(s) + CO2(g) Group of answer choices A positive B negative C zeroarrow_forward
- Which reaction is most likely to have a positive ΔSsys?a) SiO2(s) + 3 C(s)¡SiC(s) + 2 CO( g)b) 6 CO2( g) + 6 H2O( g)¡C6H12O6(s) + 6 O2( g)c) CO( g) + Cl2( g)¡COCl2( g)d) 3 NO2( g) + H2O(l )¡2 HNO3(l ) + NO( g)arrow_forwardFor the reaction below, which statement best describes what is happening with the sign of ΔG0? N a2C O3(a q) + C a Cl2(a q) → C a C O3(s) + 2 N a Cl(a q) (endothermic) Question 6 options: The reaction is non-spontaneous at low temperature and spontaneous at high temperature. The reaction is spontaneous at all temperatures. The reaction is spontaneous at low temperature and non-spontaneous at high temperature. The reaction is non-spontaneous at all temperatures.arrow_forwardThe following questions refer to the following reaction at constant 25°C and 1 atm.2Fe(s) + (3/2)O2(g) + 3H2O(l) 2Fe(OH)3(s) H = –789 kJ/molSubstance S° (J/mol K)Fe(OH)3(s) 107Fe(s) 27O2(g) 205H2O(l) 70a. Determine Ssurr for the reaction (in kJ/mol K)b. Determine Suniv for the reaction (in kJ/mol K)2.65c. What is the G for this reaction?d. Calculate the equilibrium constantarrow_forward
- Give typed explanation Under which set of conditions is ΔG°rxn for the reaction A(g) → B(g) most likely to be negative? a) PA = 10.0 atm; PB = 10.0 atm b) PA = 10.0 atm; PB = 0.010 atm c) PA = 0.010 atm; PB = 10.0 atm d) PA = 0.010 atm; PB = 0.010 atmarrow_forwardConsider the following thermochemical equations. Reaction ΔrH° / kJ mol−1 HBr(g) ⟶ H(g) + Br(g) 365.7 H2(g) ⟶ 2 H(g) 436.0 Br2(g) ⟶ 2 Br(g) 193.9 What is ΔrH° for the reaction below? HBr(g) ⟶ ½ H2(g) + ½Br2(g) Give your answer in kJ mol−1, accurate to one decimal place. Kindly double check your solution.arrow_forwardFor each process below, determine the sign of ΔG. (positive, negative, or not enough information)2H2O(s) → 2H2O(l) at relatively high temperaturesA process for which ΔS is negative.2H2(g) + O2(g) → 2H2O(g) ΔH= -An exothermic process during which the atoms get more ordered.A process with positive ΔH and positive ΔS.A process that is non-spontaneous at all temperatures.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning


