
Concept explainers
(a)
Interpretation : Need to comprehend the amino acid on the basis of given R-group and also identify the type of interaction occur between these R-groups.
Concept Introduction : Amino acid contains both −COOH and − NH2 groups bonded on same C atom of the molecule. Hence the general formula of amino acid can be shown as given below.
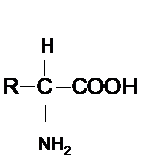
Here R-group varies for different amino acids.
(b)
Interpretation : Need to comprehend the amino acid on the basis of given R-group and also identify the type of interaction occur between these R-groups.
Concept Introduction : Amino acid contains both −COOH and − NH2 groups bonded on same C atom of the molecule. Hence the general formula of amino acid can be shown as given below.
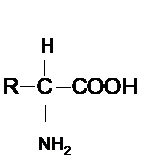
Here R-group varies for different amino acids.
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Basic Chemistry
- Draw the predominant form the glutamine amino acids at physiological pH (7.4):arrow_forwardC. Types of Steroid Hormones by Structure Steroid Hormone # of Carbon atom Structure 1. 2. 3arrow_forwardPlease explain 4. Determine the structure of compounds A-F in the following reaction şequence.(17.43) CI Br 1. СН3 (еxcess) KCN A 1. LIAIH,, ether В 2. H,О HO, PBrg 2. NaOH, A 1. NaH NaBH, 1. NaH F 2. CH,CH2Br 2. CH,CH,Br CH,CH2OHarrow_forward
- Draw the predominant form for each of the following amino acids at physiological pH (7.4): a. aspartate b. Histidine c. argininearrow_forward7. 8. 9. A Br Provide IUPAC name: 10. Arginine at pH=7 Draw structure OH leq H₂SO4 OH Provide missing reagent leq H₂SO4 KOH H₂O Enough base to raise pH ΝΗ IO Name the reaction: Provide product structure Name functional group Arginine at pH = 11 Draw the product(s) Draw structurearrow_forwardWhat type of bond holds these two monosaccharides together in this structure? HO- H. -O H H. OH A H HO 3. 12 HO. HB HO 3 1 HO OH ) B(1,4) glycosidic bond 2 a (1,4) glycosidic bond 3 aB (1.4) glycosidic bond 4) a (1,6) glycosidic bond INarrow_forward
- 15.6 Stearic acid and linoleic acid each have 18 carbon atoms. Why does stearic acid melt at 69 °C but linoleic acid melts at -5 °C? 15.7 Draw the line-angle formula for each of the following fatty acids: a. palmitic acid b. oleic acid 15.8 Draw the line-angle formula for each of the following fatty acids: a. stearic acid b. linoleic acidarrow_forwardWhich functional group on furosemide’s structure below is most likely to be negatively charged at physiological pH (7.4)?arrow_forward16.6 Draw the structure for each of the following amino acids at physiological pH: a. lysine d. Tyr 16.7 Classify each of the amino acids in problem 16.5 as polar or nonpolar. If polar, indicate if the R group is neutral, acidic, or basic. Indicate if each is hydrophobic or hydrophilic. b. proline c. V 16.8 Classify each of the amino acids in problem 16.6 as polar or nonpolar. If polar, indicate if the R group is neutral, acidic, or basic. Indicate if each is hydrophobic or hydrophilic. Cibaarrow_forward
- Activity: concept Map Directions: Use the terms below to create a concept map. Amino Acids Lipids Proteins Carbohydrates monosaccharides RNA catalysts Nucleic Acids store genetic information main energy source Nucleotides long-term energy storage Fats polysaccharides serve as transporters BIOMOLECULES 3. 4. whose main function is whose main function is which are made of which are made of 8. 7. whose main function is which include whose many functions include disaccharide oils 14. muscle struct ure 1. which include 10. 12. DNA 13. 15.arrow_forward(19, 9) Draw a structural formula for the major organic product of the reactions shown below. You do not have to consider stereochemistry.arrow_forward15.34 Use line-angle formulas to write the balanced chemical equation for the acid hydrolysis of glyceryl trioleate (triolein). 15.35 Use condensed structural formularrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





