
Interpretation:
The structure of hydrogen chloride salt of promazine has to be drawn.
Concept Introduction:
Promazine: It is a type of drug used as an antipsychotic tranquilizer. It is consumed as hydrogen chloride salt of promazine.
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
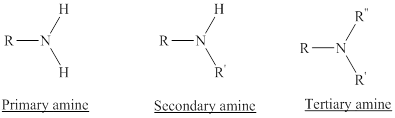
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
In a quaternary ammonium ion a nitrogen atom with four attached groups is positively charged. And their compounds are known as quaternary ammonium salt.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- The specific gravity of the following compounds are: Ether – 0.715; Bromine - 3.016; Glycerin – 1.25; and Chloroform - 1.476. If 22 ml of the unknown solution weighs 32.47 g, what is the identity of the solution? Ether None of the choices Bromine Chloroform Glycerinarrow_forwardGive the formulas for the ions in the following two ionic compoundscation (+) anion (-)ammonium chloride _________ _________sodium chloride _________ _________The bromide ion (made from bromine atoms) has the same charge as the chloride ion. Writethe chemical formula for the following two ionic compounds where the bromide ion issubstituted for the chloride ion.ammonium bromide = ________________; sodium bromide =____________________arrow_forwardHow many mL of methadone is needed for a patient who was prescribed 0.015 g of methadone? The vial reads 6 milligrams of methadone per 1 mL of solution.arrow_forward
- Demerol 25 mg IM is ordered stat. On hand is Demerol 100 mg per 2 ml. How many ml would you give?arrow_forwardWrite the chemical formula for the conjugate base of each of the following acids: Carbonic Acid (H2CO3)___________________ Formic Acid (CH2O2)_____________________arrow_forwardWrite the chemical formula of the conjugate base of boric acid.arrow_forward
- The density of trifluoroacetic acid vapor was determined at 118.1 °C and 468.5 torr, and found to be 2.784 g/L. Calculate K, for the association of the acid. 0-H...O 2CF,CO,H(g) = CF;C CF,C(g) O----H-Oarrow_forwardDetermine the pH for the following solutions. {H3O+}=2.4x10^-2 Marrow_forwardWhat is diazepam (Valium)?arrow_forward
- How many grams of NaOH would you need to make 3,982 mL of a 3 M NAOH solution (m.w. = 40.00 g/mol)? Report your answer in the requested units to the nearest 2 decimal places (Example "40.02"). Do not report the units in your answer; enter the numbers only. It is recommended that you do not round between calculation steps.arrow_forwardYou are asked to prepare 50 ml of a 1:100 rifampin (rifadin) and you have in stock a 1:20 rifampin suspension. How many milliliter of the 1:20 suspension and how many milliliter of water will you need?arrow_forwardThe following compound can be described as a(an)arrow_forward
