
Concept explainers
Interpretation:
Four examples for beta-lactam antibiotics have to be drawn. The
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Ester: One
Amide: One
Alkene: Unsaturated hydrocarbons having atleast one double bond between two carbon atoms are known as alkenes.
Ether: Ether is a group of organic compound where two aryl or alkyl groups are connected by an oxygen atom. It is represented as
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
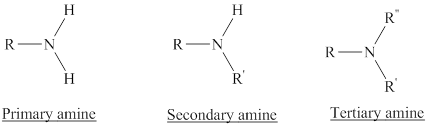
Lactam: They are cyclic amides where the carbonyl carbon part of carboxylic acid group and nitrogen part of amine group of the same compound attached forming a ring. Beta-lactam is a membered ring with two carbon atoms.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Please Provide the name of the following nitrogen-containing heterocyclic base for both of the followingarrow_forwardMethionine is an essential amino acid that can be metabolized to homocysteine. Using the figure below, describe the R-group on homocysteine and how it differs from methionine. Include the functionality of the thiol as part of this discussion.arrow_forwardFor each of the following amino acids: 1) describe what the important functional group of the amino acid side chain is, 2) identify the charge of the side chain, 3) name another component in the structure that the amino acid might interact with, and 4) indicate how the interaction with the component listed in 3) might be occurring (i.e., electrostatic interaction, hydrogen bond, hydrophobic interaction, etc.) a) Asp153 b) Arg166 c) Lys167arrow_forward
- What are the donor atoms involved in Aspartame-Cu(II) binding? A) Nitrogen atom of alpha amine group of Aspartic acid and an oxygen atom of alpha carboxyilic acid group of Aspartic acid B) Nitrogen atom of alpha amine group of Phenyl alanine methyl ester and an oxygen atom of alpha carboxyilic acid group of Aspartic acid C) Nitrogen atom of alpha amine group of Aspartic acid and an oxygen atom of alpha carboxyilic acid group of Phenyl alanine methyl ester D) Oxygen atoms of the dipeptide Aspartame E) Nitrogen atom of alpha amine group of Aspartic acid and an oxygen atom of carboxyilic acid group of the R group of Aspartic acidarrow_forwardThe compound shown here is used to treat trypanosome infection. What amino acid does the compound resemble?arrow_forwardThe cofactor shown below: is an oxidizing agent is a reducing agent is a carrier of acyl groups is flavin mononucleotide The cofactor shown below: CH2 HC OH HC OH HC OH CH2 H₂C NH H H H OH OH NH₂ 1) is an oxidizing agent 2) is a reducing agent 3) is a carrier of acyl groups 4) is flavin mononucleotidearrow_forward
- a) Write out the structure of the cofactor required for each of the following reactions. b) For each cofactor, give a specific (i.e. no generic substituents such as X or R) example of a reaction requiring the cofactor. a) Methylation. b) Amino acid decarboxylation. c) A Claisen reaction. d) Ketoacid decarboxylation. e) Aldehyde reductionarrow_forwardAfter a polypeptide chain has been synthesized, certain amino acids in the peptide may become modified. For each modified amino acid, identify the standard amino acid from which it is derived. Enter the unabbreviated name of the standard amino acid. OH I *NH,CH,–C–CH,CH,—CH–COO H C *NH₂ standard amino acid: Identify the modification that yields the first amino acid depicted. addition of a hydroxyl group addition of an amino group modification of the existing amino group modification of the existing hydroxyl group elongation of the side chain by the addition of a -CH₂- grouparrow_forwardWhen D-gulose forms a ring structure, a) b) a ketone and a hydroxyl group react to form a hemiketal. the ring is unstable at neutral pH. the ring contains four stereogenic centers. an intramolecular reaction creates a glycosidic bond.arrow_forward
- Make a table consisting of the following classes of Amino Acids (A). Give the 3 letter abbreviation, one letter symbol. Classify if the AA is essential or non essential, polar or non-polar. Write A for essential and B for non essential, write P for for polar and N for non polar. Draw the AA in the ff. format: Encircle the -R (side chain)do not include met in aliphaticamino acids. it has a separate box inS containing amino acidsarrow_forwardAn octapeptide was hydrolyzed and the amino acids separated. Its amino acid composition was determined: Gly (1); Ala (2); Val (1); Lys (2); Met (1); Asp (1). After incubation with FDNB and hydrolysis, 2,4-dinitrophenylalanine was detected. NO, H N -C- COOH H O,N CH, Treatment with cyanogen bromide, which cleaves at M, resulted in two fragments: a pentapeptide containing two K residues and a tripeptide containing V, G, and A. The residues are not listed in any particular order. After treatment with trypsin, which cleaves at R and K, there were three fragments: two dipeptides and MGAV. What is the sequence of the octapeptide? Use the one-letter abbreviations for the amino acids, and do not use dashes. octapeptide sequence:arrow_forwardThe following structures were introduced as neuromuscular blocking agents. Structure B is derived from structure A. What was the main reason for incorporating additional ester groups? O To enhance hydrogen bonding interactions with the binding site. O To increase the polarity of the drug. O To decrease the stability of the drug O To alter the conformation of the molecule.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





