
(a)
Interpretation:
The name and structure of three different amides with formula
Concept Introduction:
Amide: One
Depending on the number of carbon side chain of the amide, different types of amides can form.
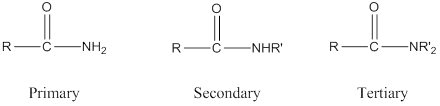
Primary amides can be named in the IUPAC system in several ways,
For simple amides the suffix – amide is added to the name of the alkyl substituent.
The suffix- amide can be used in place of the final –e in the name of the parent compound.
For a secondary amides an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amides an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Ammonium salt is formed when tertiary
(b)
Interpretation:
The name and structure of three different esters with formula
Concept Introduction:
In chemistry structure is the arrangement of
According to
The order of priority is,
Carboxylic acid: One
Ester: One
In case of carboxylic esters “–oic acid”is replaced by “esters “-oate” is to be written. The first part of the name indicates the alkyl group in the
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Draw condensed structural formulas for the two carboxylic acids with the molecular formula C4H8O2arrow_forwardExamine the structures of the following molecules. In each compound circle and identify by name each functional group. a) ethanolamine b) Glucosamine c) Threonine H₂N-C-C-OH H₂ H₂ H₂N HO OH d) Phosphoenolpyruvate с H₂N-CH OH OH OH HC - OH 1 CH3 OH T O=P-O O H₂C=C-C OHarrow_forwardA) Describe the glycosidic bond (using standard convention) indicated by “Arrow a.” B) Draw the open chain Fischer projection formula of the monosaccharide labeled “B” C) Describe the glycosidic bond (as in question A) indicated by “Arrow b.”arrow_forward
- Identify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2arrow_forwardProvide compounds that fit the following descriptions:(a) Two amines that are gases at room temperature(b) A heterocyclic amine(c) A compound with an amine group on an aromatic ringarrow_forwardDraw the structures of the following compounds. (Includes both new and old names.) 3-cyclopentylhexan-3-olarrow_forward
- Draw structures corresponding to the following names:(a) 3-Methylhexan-1-ol (b) 1-Methyl-3-propylcyclopentanol(c) 2,2-Dimethylhexan-3-ol (d) Heptan-3-ol(e) 2,3-Diethylcyclohexanolarrow_forwardSpermaceti, a fragrant substance isolated from sperm whales, was commonly used in cosmetics until it was banned in 1976 to protect the whales from extinction. Chemically, spermaceti is cetyl palmitate, the ester of palmiticacid with cetyl alcohol (the straight-chain 16-carbon alcohol). Draw the structure of spermaceti.arrow_forwardWhich of the following define the stereochemistry of alanine (as per the structure shown)? Note: Functional groups arranged horizontally are facing towards the front, and the functional groups arranged vertically are facing towards the back. СООН + H₂N to OS- Od- CH, OR-arrow_forward
- Histidine has three ionizable groups. On the titration curve below, show: a) 2 molar equivalents of OH b) pka of the third ionizable group Histidine Titration 12.0 10.0 8.0 4 pH 6.0 4.0 2.0 0.0 Equivalents OHarrow_forwardCompound A undergoes a reaction with hydrogen bromide, HBr to produce2-bromobutane. A exists as cis-trans isomers and decolourises brominesolution in methylene chloride, CH2Cl2. a)Draw and name the structure of compound D. b)Draw two (2) constitutional isomers of compound Darrow_forward(a) Draw the condensed structural formula, and give the name and abbreviation for the dipeptide Cys-Pro. (b) For the sugar with the structure shown below: (i) draw an arrow pointing to the anomeric carbon (ii) Is this the alpha or Beta anomer? ОН H OH OH OH OHarrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





