
Physics (5th Edition)
5th Edition
ISBN: 9780321976444
Author: James S. Walker
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17, Problem 45PCE
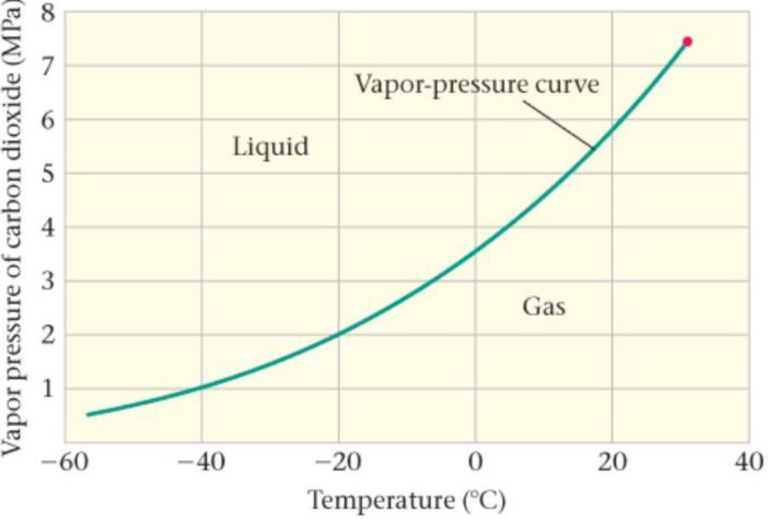
Predict/Calculate The Vapor Pressure of CO2 A portion of the vapor-pressure curve for carbon dioxide is given in Figure 17-36. (a) Estimate the pressure at which CO2 boils at 0 °C. (b) If the temperature is increased, does the boiling pressure increase, decrease, or stay the same? Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a) Estimate the average spacing between the molecules of 1 mol of an ideal gas at a pressure of 1atm and a temperature of 300 K. b) 1 mol of liquid water occupies a volume of 18 cm. Estimate the spacing between molecules. c) Use your result from part (a) to estimate the diameter of a water molecule. d) Estimate the factor by which water expands when it boils
8 of 13
I Review I Constants
Oxygen (O2) has a molar mass of 32.0 g/mol .
Part A
What is the root-mean-square speed of an oxygen
molecule at a temperature of 293 K ?
Express your answer in meters per second.
ΑΣφ
?
Vrms
Submit
Request Answer
Part B
What is its average translational kinetic energy at that
speed?
Express your answer in joules.
PERUUS
A metal has an initial oxide coating thickness, y, of 60 nm. After 2 hours at 700°C
the oxide has grown to a thickness of 80 nm.
Assuming a parabolic growth rate law applies, i.e. y² = c t + d (where, t is the time in
hours and both c and d are constants) what will be the approximate thickness of the
coating after a total of 36 hours at 700°C?
210 nm.
116 nm.
O 232 nm.
420 nm.
126 nm.
SVOJE VŠET
Chapter 17 Solutions
Physics (5th Edition)
Ch. 17.1 - Rank the following ideal-gas systems in order of...Ch. 17.2 - If the Kelvin temperature of a gas is doubled, by...Ch. 17.3 - A metal rod of a given initial length and...Ch. 17.4 - A portion of a substances phase diagram is shown...Ch. 17.5 - Which requires more heat: melting 100 kg of copper...Ch. 17.6 - An ice cube is placed in a cup of water. A few...Ch. 17 - How is the air pressure in a tightly sealed house...Ch. 17 - The average speed of air molecules in your room is...Ch. 17 - Is it possible to change both the pressure and the...Ch. 17 - Prob. 4CQ
Ch. 17 - A camping stove just barely boils water on a...Ch. 17 - An autoclave is a device used to sterilize medical...Ch. 17 - As the temperature of ice is increased, it changes...Ch. 17 - BIO Isopropyl alcohol is sometimes rubbed onto a...Ch. 17 - A drop of water on a kitchen counter evaporates in...Ch. 17 - (a) Is the number of molecules in one mole of N2...Ch. 17 - Predict/Explain If you put a helium-filled balloon...Ch. 17 - Two containers hold ideal gases at the same...Ch. 17 - Prob. 4PCECh. 17 - BIO After emptying her lungs, a person inhales 4.3...Ch. 17 - An automobile tire has a volume of 0.0185 m3. At a...Ch. 17 - Prob. 7PCECh. 17 - A compressed-air tank holds 0.500 m3 of air at a...Ch. 17 - Four ideal gases have the following pressures, P,...Ch. 17 - A balloon contains 3.9 liters of nitrogen gas at a...Ch. 17 - Prob. 11PCECh. 17 - Predict/Calculate A bicycle tire with a volume of...Ch. 17 - A 515-cm3 flask contains 0.460 g of a gas at a...Ch. 17 - Prob. 14PCECh. 17 - The air inside a hot-air balloon has an average...Ch. 17 - Prob. 16PCECh. 17 - Consider the system described in the previous...Ch. 17 - Prob. 18PCECh. 17 - Prob. 19PCECh. 17 - If the translational speed of molecules in an...Ch. 17 - At what temperature is the rms speed of H2 equal...Ch. 17 - Suppose a planet has an atmosphere of pure ammonia...Ch. 17 - Prob. 23PCECh. 17 - Prob. 24PCECh. 17 - Prob. 25PCECh. 17 - What is the temperature of a gas of CO2 molecules...Ch. 17 - The rms speed of a sample of gas is increased by...Ch. 17 - Prob. 28PCECh. 17 - A 380-mL spherical flask contains 0.065 mol of an...Ch. 17 - Prob. 30PCECh. 17 - A rock climber hangs freely from a nylon rope that...Ch. 17 - BIO To stretch a relaxed biceps muscle 2.5 cm...Ch. 17 - A 22-kg chimpanzee hangs from the end of a...Ch. 17 - The Marianas Trench The deepest place in all the...Ch. 17 - Four cylindrical rods with various cross-sectional...Ch. 17 - Predict/Calculate A steel wire 4.1 m long...Ch. 17 - BIO Spiderweb An orb weaver spider with a mass of...Ch. 17 - Predict/Calculate Two rods of equal length (0.55...Ch. 17 - A piano wire 0.82 m long and 0.93 mm in diameter...Ch. 17 - The formation of ice from water is accompanied by...Ch. 17 - Vapor Pressure for Water Figure 17-35 shows a...Ch. 17 - Using the vapor-pressure curve given in Figure...Ch. 17 - Prob. 43PCECh. 17 - Prob. 44PCECh. 17 - Predict/Calculate The Vapor Pressure of CO2 A...Ch. 17 - Phase Diagram for Water The phase diagram for...Ch. 17 - Phase Diagram for CO2 The phase diagram for CO2 is...Ch. 17 - Prob. 48PCECh. 17 - How much heat must be removed from 1.96 kg of...Ch. 17 - A heat transfer of 9.5 105 J is required to...Ch. 17 - How much heat must be added to 2.55 kg of copper...Ch. 17 - An ammonia refrigeration cycle involves the...Ch. 17 - Prob. 53PCECh. 17 - Prob. 54PCECh. 17 - Prob. 55PCECh. 17 - Figure 17-30 shows a temperature-versus-heat plot...Ch. 17 - Predict/Calculate Suppose the 1.000 kg of water in...Ch. 17 - Prob. 58PCECh. 17 - When you go out to your car one cold winter...Ch. 17 - A large punch bowl holds 3.99 kg of lemonade...Ch. 17 - A 155-g aluminum cylinder is removed from a liquid...Ch. 17 - An 825-g iron block is heated to 352 C and placed...Ch. 17 - Party Planning You are expecting to serve 32 cups...Ch. 17 - Predict/Calculate A 35-g ice cube at 0.0 C is...Ch. 17 - A 48-g block of copper at 12 C is added to 110 g...Ch. 17 - A 0 075-kg ice cube at 0.0 C is dropped into a...Ch. 17 - To help keep her barn warm on cold days, a farmer...Ch. 17 - CE As you go up in attitude, do you expect the...Ch. 17 - Prob. 69GPCh. 17 - Prob. 70GPCh. 17 - Prob. 71GPCh. 17 - Cooling Computers Researchers are developing heat...Ch. 17 - Prob. 73GPCh. 17 - Prob. 74GPCh. 17 - Evaporating Atmosphere Hydrogen gas evaporates...Ch. 17 - Prob. 76GPCh. 17 - A Boiling Geyser (a) The column of water that...Ch. 17 - A Melting Glacier (a) A glacier is made of ice of...Ch. 17 - Peter catches a 4 2-kg striped bass on a fishing...Ch. 17 - A steel ball (density=7860kg/m3) with a diameter...Ch. 17 - A lead brick with the dimensions shown in Figure...Ch. 17 - (a) Find the amount of heat that must be extracted...Ch. 17 - Mighty Ice Lift A tremendous force is generated...Ch. 17 - Orthopedic Implants Metals such as titanium and...Ch. 17 - Students on a spring break picnic bring a cooler...Ch. 17 - A 5.9-kg block of ice at 1.5 C slides on a...Ch. 17 - A cylindrical copper rod 37 cm long and 7.5 cm in...Ch. 17 - Prob. 88PPCh. 17 - Prob. 89PPCh. 17 - Prob. 90PPCh. 17 - Prob. 91PPCh. 17 - Referring to Example 17-17 (a) Find the final...Ch. 17 - Referring to Example 17-17 (a) Find the final...
Additional Science Textbook Solutions
Find more solutions based on key concepts
31. A weather balloon rises through the atmosphere, its volume expanding from 4.0 m3 to 12 m3 as the temperatur...
College Physics: A Strategic Approach (3rd Edition)
The height of a certain hill (in feet) is given by , where y is the distance (in miles) north, x the distance e...
Introduction to Electrodynamics
36.51 Measuring Refractive Index. A thin slit illuminated by light of frequency f produces its first dark band ...
University Physics with Modern Physics (14th Edition)
In this activity, we will use a representation of the atom in which a central nucleus containing the protons an...
Lecture- Tutorials for Introductory Astronomy
18. The distance between the objective and eyepiece of a telescope is 55 cm. The focal length of the eyepiece i...
College Physics: A Strategic Approach (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- (a) If tossing 100 coins, how many ways (microstates) are there to get me three most likely macro states of 49 heads and 51 tails, 50 heads and 50 tails, and 51 heads and 49 tails? (b) What percent of the total possibilities is this? (Consult Table 15.4.)arrow_forward(a) Hydrogen molecules (molar mass is equal to 2.016 g/mol) have vrms equal to 193 m/s. What is the temperature? (b) Much of the gas near the Sun is atomic hydrogen (H rather than H2). Its temperature would have to be 1.5107 K for the speed vrms to equal the escape velocity from the Sun. What is that velocity?arrow_forward(a) What is me vapor pressure of water at 20.0C ? (b) What percentage of atmospheric pressure does this correspond to? (c) What percent of 20.0C air is water vapor if it has 100% relative humidity? (The density of dry air at 20.0C is 1.20kg/m3 .)arrow_forward
- (a) Construct a table showing the macro states and all of the individual microstates for tossing 13 coins. (Use Table 15.5 as a guide.) (b) How many macro states are there? (c) What is the total number of microstates? (d) What percent chance is here of tossing 5 heads and 1 tail? (e) How much more likely are you to toss 3 heads and 3 tails than 5 heads and 1 tail? (Take me ratio of the number of microstates to find out.)arrow_forwardRelative Humidity One measure of the amount of water in the atmosphere is the relative humidity, the ratio of the actual partial pressure of water vapor to the maximum possible partial pressure of water. If the partial pressure of water vapor exceeds the vapor pressure ( Figure 1) at that temperature, the vapor condenses to liquid and either fog or dew begins to form. In that case the relative humidity would be 100%. Figure Vapor pressure of water (kPa) 8 O 10 Liquid Vapor-pressure curve 20 Temperature (°C) Gas 30 40 1 of 1 Part A Referring to (Figure 1), if the partial pressure of water in the atmosphere is 1.7 kPa when the air temperature is 25°C, what is the relative humidity? Express your answer to two significant figures and include appropriate units. 0.54 Submit μA Provide Feedback Units ? Previous Answers Request Answerarrow_forwardWithout completing the calculations, determine what the new pressure will be in a problem below. Also, explain how you were able to determine the new pressure without completing the calculations . A sample of ozone (O3) is stored at x kelvin and 12.5 Pa . If the temperature is doubled 2x kelvin, what will the new pressure be?arrow_forward
- Q7: Hydrogen gas filled balloons are used for survey of weather at higher altitudes. Such hydrogenballoon at Earth’s surface has a volume of 5000 mm3 on a day when the temperature is 27 ° C and the pressure is 1MPa. The balloon rises and expands as the pressure drops. What would the volume of thesame number of moles of hydrogen be at an altitude of 40 km where the pressure is 0.33 MPa and thetemperature is −13 ° C? If we travel from ground to sky, atmospheric pressure changes? Assess it andjustify your result.arrow_forward8. At 20°C (approximately room temperature), the average velocity of N2 molecules in air is approximately 469m/s. What is the kinetic energy of an Na molecule moving at this speed? 9. A tank volume of 0.3m' contains 2 mol of He gas at 20°C. Assuming an ideal gas, calculate: (a) The total thermal energy of the system (b) The average kinetic energy per moleculearrow_forward7. If I have an unknown quantity of gas held at a temperature of 1195 K in a container with a volume of 25 liters and a pressure of 560 atm, how many moles of a. 143.0mol gas do I have? d. 0.3457mol b. 0.01004mol C. 146.0mol 8. If I have 21 males of g3s held at aarrow_forward
- 1. on a hot summer day the temperature is 40.0°c and the pressure is 1.01x10^5 Pa. et us model the air as all nitrogen of molecular mass 28.0g/mol having molecules of diamtere 0.500 nm (nano = 10^-9) that are moving at their root-mean-square speed. Avogadro's number is 6.02x10^23 molecules/mol, the ideal gas constant is 8.31 J/mol . K, and the boltzmann constant is 1.38x10^-23 J/K calculate reasonable estimates for A. the root mean-square speed of the nitrogen molecules B. the average distance of a typical molecule travels between collisions C. the average time a molecule travels between collisions, assuming that the molecules are moving at their root-mean-swuare speeds D. the molecule of collisions an average molecule undergoes per second 2. A 648-G Empty Iron Kettle Is Put On A Stove. How Much Heat, In Joules, Must It Absorb To Raise Its Temperature From 15.0°C To raise its temperature from 15°c to 37°c (the specific heat for iron is 113 ca;/g°C, 1 cal = 9.190J)arrow_forwardQUESTION 8 What is the density in kg/m° of diatomic oxygen gas, 02, that is under a pressure of 100 kPa at a temperature of 280 K? The atomic weight of the element oxygen is 16 g/mole. а. 0.20 kg/m3 b. 1.1 kg/m3 C. 0.50 kg/m3 d. 0.80 kg/m3 e. 1.4 kg/m3arrow_forward3. Solid molecular hydrogen. For H₂ one finds from measurements on the gas that the Lennard-Jones parameters are & = 50×10-¹6 erg_and o = 2.96 Å. Find the cohesive energy in kJ per mole of H₂; do the calculation for an fcc structure. Treat each H₂ molecule as a sphere. The observed value of the cohesive energy is 0.751 kJ/mol., much less than we calculated, so that quantum corrections must be very important.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY