
Basic Chemistry
6th Edition
ISBN: 9780134878119
Author: Timberlake, Karen C. , William
Publisher: Pearson,
expand_more
expand_more
format_list_bulleted
Question
Chapter 16.1, Problem 5PP
Interpretation Introduction
To determine:Identification of the following
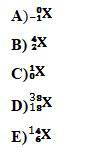
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
8. The following question is from my textbook practice questions. It is shown in the photo below.
55R) The density of lead is 11.4 g/cm3. The mass of a lead ball with a radius of 0.50 mm is
_g.
4nr.
(n = 3.1416; Vsphere = "n3/3)
A) 6.0
B) 4.6 x 10-2
C) 4.6 x 10-5
D) 6.0 x 10-3
E) 4.6
proposed
multiplication or division
Is this
possible?
result
10. g
?
yes
5.0 L
no
(s.0 m²) - (s.0 m) = ?
3
O yes
no
(20 ) - (0.050 kg) – ?
yes
2.0 g´ ) ·
(0.050 kg) = ?
no
Chapter 16 Solutions
Basic Chemistry
Ch. 16.1 - Prob. 1PPCh. 16.1 - Prob. 2PPCh. 16.1 - Naturally occurring potassium consists of three...Ch. 16.1 - Prob. 4PPCh. 16.1 - Prob. 5PPCh. 16.1 - Prob. 6PPCh. 16.1 - Prob. 7PPCh. 16.1 - Prob. 8PPCh. 16.1 - Supply the missing information in the following...Ch. 16.1 - Prob. 10PP
Ch. 16.1 - Prob. 11PPCh. 16.1 - Prob. 12PPCh. 16.2 - Prob. 13PPCh. 16.2 - Prob. 14PPCh. 16.2 - Prob. 15PPCh. 16.2 - Prob. 16PPCh. 16.2 - Prob. 17PPCh. 16.2 - Prob. 18PPCh. 16.2 - Prob. 19PPCh. 16.2 - Prob. 20PPCh. 16.2 - Prob. 21PPCh. 16.2 - Prob. 22PPCh. 16.3 - Prob. 23PPCh. 16.3 - Prob. 24PPCh. 16.3 - Prob. 25PPCh. 16.3 - Prob. 26PPCh. 16.3 - Prob. 27PPCh. 16.3 - Prob. 28PPCh. 16.4 - Prob. 29PPCh. 16.4 - Prob. 30PPCh. 16.4 - Prob. 31PPCh. 16.4 - Prob. 32PPCh. 16.4 - Prob. 33PPCh. 16.4 - Prob. 34PPCh. 16.5 - Prob. 35PPCh. 16.5 - Prob. 36PPCh. 16.5 - Prob. 37PPCh. 16.5 - Prob. 38PPCh. 16.5 - Prob. 39PPCh. 16.5 - Prob. 40PPCh. 16.6 - Prob. 41PPCh. 16.6 - Prob. 42PPCh. 16.6 - Prob. 43PPCh. 16.6 - Prob. 44PPCh. 16.6 - Prob. 45PPCh. 16.6 - Prob. 46PPCh. 16.6 - Prob. 47PPCh. 16.6 - Prob. 48PPCh. 16.6 - Prob. 49PPCh. 16.6 - Prob. 50PPCh. 16 - Prob. 51UTCCh. 16 - Prob. 52UTCCh. 16 - The chapter sections to review are shown in...Ch. 16 - Prob. 54UTCCh. 16 - Prob. 55UTCCh. 16 - Prob. 56UTCCh. 16 - Prob. 57APPCh. 16 - Prob. 58APPCh. 16 - Prob. 59APPCh. 16 - Prob. 60APPCh. 16 - Prob. 61APPCh. 16 - Prob. 62APPCh. 16 - Prob. 63APPCh. 16 - Prob. 64APPCh. 16 - Prob. 65APPCh. 16 - Prob. 66APPCh. 16 - Prob. 67APPCh. 16 - Prob. 68APPCh. 16 - Prob. 69APPCh. 16 - Prob. 70APPCh. 16 - Prob. 71APPCh. 16 - Prob. 72APPCh. 16 - Prob. 73APPCh. 16 - Prob. 74APPCh. 16 - Prob. 75APPCh. 16 - Prob. 76APPCh. 16 - Prob. 77APPCh. 16 - Prob. 78APPCh. 16 - Prob. 79CPCh. 16 - Prob. 80CPCh. 16 - Prob. 81CPCh. 16 - Prob. 82CPCh. 16 - Prob. 83CPCh. 16 - Prob. 84CPCh. 16 - Prob. 85CPCh. 16 - Prob. 86CPCh. 16 - Prob. 87CPCh. 16 - Prob. 88CPCh. 16 - Prob. 89CPCh. 16 - Prob. 90CPCh. 16 - Consider the reaction of sodium oxalate (Na2C2O4)...Ch. 16 - Prob. 34CICh. 16 - Prob. 35CICh. 16 - Prob. 36CICh. 16 - Prob. 37CICh. 16 - Prob. 38CICh. 16 - Prob. 39CICh. 16 - Prob. 40CI
Knowledge Booster
Similar questions
- What is the weight of Amphotec®, an antibiotic preparation for aspergillus infection, necessary for a 135 pound patient at the rate of 3.5 mg/kg per dose? (Given: 1kg = 2.205 lbs) a. 61.4 mg b. 2228 mg c. 7.7 mg d. 214 mgarrow_forwardRound each number as indicated a) 8.3158 (3 SF) b) 25225 (3 SF)arrow_forwardBromine-88 is radioactive and has a half ife of 16.3 seconds. What percentage of a sample would be left after 10.5 seconds? Round your answer to 2 significant digits. D.Parrow_forward
- The normal dosage for azinthromycin, an antibiotic, is 3.3 mg/kg. What is the proper dose for a 150 lb patient? (Given 1kg 2.205 lbs) A.100 mg B.224 mg C.300 mg D. 495 mgarrow_forwardHome work and Exercises 1.53 and 1.56 53. Convert the following Fahrenheit temperatures to the Celsius and Kelvin scales. a. -459°F, an extremely low temperature b. -40.°F, the answer to a trivia question c. 68 F, room temperature d. 7 x 10 F. temperature required to initiate fusion reactions in the sunarrow_forward7.The density of osmium is 22.6 g/cm³. A sample of osmium with a mass of 21.8 g would occupy a volume of cm3. a. 0.965 b. 1.04 c. 493 d. 2.03 x 10-3 e. 2.03 x 10-3arrow_forward
- What is the density of a rectangular solid which has a mass of 25.71 g and is 2.30 cm long, 4.01 cm wide and 1.82 cm high (in g/cm3)Watch sigfigs? Group of answer choices A. 14.5 B. 7.23 C. 1.53 D. 0.653arrow_forward2. Classify each of the following changes as primarily chemical or physical in nature: a. protein being digested by stomach enzymes c. ice melting e. blowing up a balloon b. wax melting d. wood burning f. paper burningarrow_forwardIdentify the estimated digit and the magnitude of uncertainty in each of the following measurements. a. 234 b. 234.0 c. 0.234 d. 0.00234arrow_forward
- F2 © Macmillan Learning NO₂ Incorrect # 3 20 F3 : 0: || $ 4 F4 078⁰ % 5 BeCL MacBook Air < 60 & 7arrow_forwardSome properties of X, Y, Z at constant pressure are given in following table. PROPERTY Change Not change Not change Temperature during fusion Including different kind of atoms Including different kind of molecules Which ones of the followings are correct according to the information given in table? I. X is a compound II. Y is an element III. Z is a pure substance 18 - O A) Only II O B) Only III OC) I. II and III O D) II and III O E) Only Iarrow_forwardHow many significant figures in each term : 0.001048 and 3032 ? a. 7 and 7 b. 6 and 6 c. 4 and 7 d. 6 and 7arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co