
Physics (5th Edition)
5th Edition
ISBN: 9780321976444
Author: James S. Walker
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16, Problem 50PCE
Figure 16-28 Problem 50
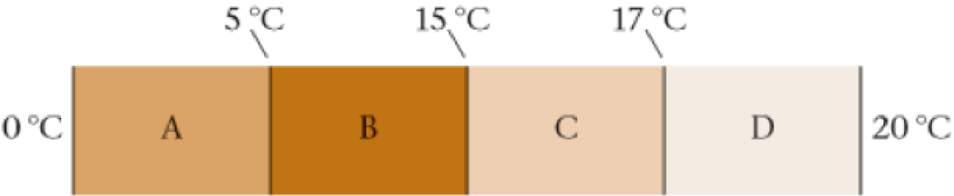
50. • CE Heat is transferred from an area where the temperature is 20 °C to an area where the temperature is 0 °C through a composite slab consisting of four different materials, each with the same thickness. The temperatures at the interface between each of the materials are given in Figure 16-28. Rank the four materials in order of increasing thermal
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
0• Yi2 49 58% 4
4G
ll 9:00 020
Example 8.4 The outside wall of a room is 4 x 3 m and 0.35 m thick. What is the
energy loss per hour if the inside and outside temperatures are 35°C and -40°C
respectively? Assume the conductivity of the wall is 0.13 W/mK.
()
II
Calculate the heat loss per m' of surface area for an
insulating wall composed of 25.4-mm-thick fiber
insulating board, where the inside temperature is
352.7 K and the outside temperature is 297.1 K.
• Thermal conductivity of fiber insulating board is
0.048-
Ax = 25.4 mm = 0.0254 m
To = 352.7 K,T, = 297.1 K
• It determines whether a certain material is a
good heat conductor or insulator.
• Question: In order to for a material to be good
conductor, what must be the value of its
thermal conductivity?
Chapter 16 Solutions
Physics (5th Edition)
Ch. 16.1 - Prob. 1EYUCh. 16.2 - Is the size of a degree in the Fahrenheit scale...Ch. 16.3 - The following systems consist of a metal rod with...Ch. 16.4 - Prob. 4EYUCh. 16.5 - Prob. 5EYUCh. 16.6 - The following systems consist of a cylindrical...Ch. 16 - Prob. 1CQCh. 16 - Prob. 2CQCh. 16 - Prob. 3CQCh. 16 - If the glass in a glass thermometer had the same...
Ch. 16 - Prob. 5CQCh. 16 - Sometimes the metal lid on a glass jar has been...Ch. 16 - Prob. 7CQCh. 16 - The specific heat of concrete is greater than that...Ch. 16 - When you touch a piece of metal and a piece of...Ch. 16 - The rate of heat flow through a slab does not...Ch. 16 - Prob. 11CQCh. 16 - Updrafts of air allow hawks and eagles to glide...Ch. 16 - BIO The fur of polar bears consists of hollow...Ch. 16 - Object 2 has twice the emissivity of object 1,...Ch. 16 - Prob. 1PCECh. 16 - Prob. 2PCECh. 16 - Incandescent lightbulbs heat a tungsten filament...Ch. 16 - Normal body temperature for humans is 98.6 F. What...Ch. 16 - The temperature at the surface of the Sun is about...Ch. 16 - One day you notice that the outside temperature...Ch. 16 - The gas in a constant-volume gas thermometer has a...Ch. 16 - Prob. 8PCECh. 16 - Greatest Change in Temperature A world record for...Ch. 16 - Prob. 10PCECh. 16 - Prob. 11PCECh. 16 - When the bulb of a constant-volume gas thermometer...Ch. 16 - Bimetallic strip A is made of copper and steel;...Ch. 16 - Prob. 14PCECh. 16 - Predict/Explain A brass plate has a circular hole...Ch. 16 - Figure 16-25 shows five metal plates, all at the...Ch. 16 - Longest Suspension Bridge The worlds longest...Ch. 16 - A vinyl siding panel for a house is installed on a...Ch. 16 - A cylinder bore in an aluminum engine block has a...Ch. 16 - Prob. 20PCECh. 16 - At 18.75 C a brass sleeve has an inside diameter...Ch. 16 - Early in the morning, when the temperature is 5.5...Ch. 16 - Some cookware has a stainless steel interior ( =...Ch. 16 - Predict/Calculate You construct two wire-frame...Ch. 16 - A metal ball that is 1.2 m in diameter expands by...Ch. 16 - A copper ball with a radius of 1.7 cm is heated...Ch. 16 - Predict/Calculate An aluminum saucepan with a...Ch. 16 - Prob. 28PCECh. 16 - BIO An exercise machine indicates that you have...Ch. 16 - BIO A certain sandwich cookie contains 53 C of...Ch. 16 - BIO During a workout, a person repeatedly lifts a...Ch. 16 - Prob. 32PCECh. 16 - BIO It was shown in Example 16-18 that a typical...Ch. 16 - Predict/Explain Two objects are made of the same...Ch. 16 - Prob. 35PCECh. 16 - Prob. 36PCECh. 16 - Prob. 37PCECh. 16 - A 9.7-g lead bullet is fired into a fence post....Ch. 16 - Prob. 39PCECh. 16 - Prob. 40PCECh. 16 - A 225-g lead ball at a temperature of 81.2 C is...Ch. 16 - If 2200 J of heat are added to a 190-g object, its...Ch. 16 - Chips by the Ton Tortilla chips are manufactured...Ch. 16 - Prob. 44PCECh. 16 - To determine the specific heat of an object, a...Ch. 16 - Predict/Calculate A student drops a 0.33-kg piece...Ch. 16 - Prob. 47PCECh. 16 - Predict/Explain In a popular lecture...Ch. 16 - Figure 16-27 shows a composite slab of three...Ch. 16 - Figure 16-28 Problem 50 50. CE Heat is...Ch. 16 - Predict/Explain Two identical bowls of casserole...Ch. 16 - Two bowls of soup with identical temperatures are...Ch. 16 - A glass window 0.33 cm thick measures 81 cm by 39...Ch. 16 - BIO Assuming your skin temperature is 37.2 C and...Ch. 16 - Find the heat that flows in 1.0 s through a lead...Ch. 16 - Consider a double-paned window consisting of two...Ch. 16 - Predict/Calculate Two metal rods of equal...Ch. 16 - Two cylindrical metal rodsone copper, the other...Ch. 16 - Prob. 59PCECh. 16 - Predict/Calculate Consider two cylindrical metal...Ch. 16 - A copper rod 85 cm long is used to poke a fire....Ch. 16 - Two identical objects are placed in a room at 24...Ch. 16 - A block has the dimensions L, 2L, and 3L. When one...Ch. 16 - Prob. 64GPCh. 16 - CE A copper ring stands on edge with a metal rod...Ch. 16 - CE Referring to the copper ring in the previous...Ch. 16 - Prob. 67GPCh. 16 - Making Steel Sheets In the continuous-caster...Ch. 16 - The Coldest Place in the Universe The Boomerang...Ch. 16 - BIO The Hottest Living Things From the surreal...Ch. 16 - Thermal energy is added to 180 g of water at a...Ch. 16 - Prob. 72GPCh. 16 - BIO Brain Power As you read this problem, your...Ch. 16 - BIO Brain Food Your brain consumes about 22 W of...Ch. 16 - BIO The Cricket Thermometer The rate of chirping...Ch. 16 - Predict/Calculate A pendulum consists of a large...Ch. 16 - Prob. 77GPCh. 16 - A256-kg rock sits in full sunlight on the edge of...Ch. 16 - Prob. 79GPCh. 16 - Thermal Storage Solar heating of a house is much...Ch. 16 - Pave It Over Suppose city 1 leaves an entire block...Ch. 16 - Prob. 82GPCh. 16 - You turn a crank on a device similar to that shown...Ch. 16 - Prob. 84GPCh. 16 - The Solar Constant The surface of the Sun has a...Ch. 16 - Bars of two different metals are bolted together,...Ch. 16 - A grandfather clock has a simple brass pendulum of...Ch. 16 - Prob. 88GPCh. 16 - A layer of ice has formed on a small pond. The air...Ch. 16 - A Double-Paned Window An energy-efficient...Ch. 16 - Cool Medicine In situations in which the brain is...Ch. 16 - Cool Medicine In situations in which the brain is...Ch. 16 - Cool Medicine In situations in which the brain is...Ch. 16 - Prob. 94PPCh. 16 - Referring to Example 16-12 Suppose the mass of the...Ch. 16 - Referring to Example 16-12 Suppose the initial...Ch. 16 - Prob. 97PPCh. 16 - Predict/Calculate Referring to Example 16-16...
Additional Science Textbook Solutions
Find more solutions based on key concepts
How are the motions in parts C and D similar? How do they differ? How are the graphs similar? How do they diffe...
Tutorials in Introductory Physics
The diagram shows Bob’s view of the passing of two identical spaceships. Anna’s and his own, where v=2 . The le...
Modern Physics
Describe the technique of radiometric dating, and explain how we know it is reliable. Be sure to explain what w...
Life in the Universe (4th Edition)
7. a. Can the normal force on an object be directed horizontally? If not, why not? If so, provide an example.
b...
College Physics: A Strategic Approach (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Thermography is a technique for measuring radiant heat and detecting variations in surface temperatures that may be medically, environmentally, or militarily meaningful.(a) What is the percent increase in the rate of heat transfer by radiation from a given area at a temperature of 34.0C compared with that at 33.0C, such as on a person’s skin? (b) What is the percent increase in the rate of heat transfer by radiation from a given area at a temperature of 34.0C compared with that at 20.0C, such as for warm and cool automobile hoods? Figure 14.35 Artist’s rendition of a thermograph of a patient’s upper body, showing the distribution of heat represented by different colors.arrow_forwardConstruct Your Own Problem Consider a person outdoors on a cold night. Construct a problem in which you calculate the rate of heat transfer from the person by all three heat transfer methods. Make the initial circumstances such that at rest the person will have a net heat transfer and then decide how much physical activity of a chosen type is necessary to balance the rate of heat transfer. Among the things to consider are the size of the person, type of clothing, initial metabolic rate, sky conditions, amount of water evaporated, and volume of air breathed. Of course, there are many other factors to consider and your instructor may wish to guide you in the assumptions made as well as the detail of analysis and method of presenting your results.arrow_forward..•P60 Buckminsterfullerene, Co0, is a large molecule consisting of 60 carbon atoms connected to form a hollow sphere. The diameter of a C60 molecule is about 7 x 10-10 m. It has been hypothesized that C60 molecules might be found in clouds of interstellar dust, which often contain interesting chemical compounds. The temperature of an interstellar dust cloud may be very low, around 3 K. Suppose you are planning to try to detect the presence of C60 in such a cold dust cloud by detecting photons emitted when molecules undergo transitions from one rotational energy state to another. Approximately, what is the highest-numbered rotational level from which you would expect to observe emissions? Rotational levels are I= 0,1,2,3,....arrow_forward
- • An aluminum pot contains water that is kept steadily boiling (100 °C). The bottom surface of the pot, which is 12 mm thick and 1.5x10* mm in area, is maintained at a temperature of 102°C by an electric heating umit. Find the rate at which heat is transferred through the bottonm surface. Compare this with a copper based pot.arrow_forward> The following gives the calorimetric values for water at various phases: . Latent heat of fusion for water: 3.33 E5 J/kg • Specific heat capacity for ice: 2,220 J/kg"C • Specific heat capacity for liquid water: 4,186 J/kg C A 15-g sample of ice at -20.0 °C is converted to liquid water at 15.0 °C. Using the values from the table above, calculate the heat required for that conversion O 4,500 J O 6,600 J O 7,800 J O 9,500 Jarrow_forward• 6) • A body skin at 38°C has a mass of 60g. When it is immersed in a current of steam at 100°C, 0.5g of steam is condensed on it. Determine the specific heat of the skin , given that the latent heat of steam = 540 cal/g. Ans. • A- 0.0726 „ft. • B- 0.0726 J/gm °C • C-0.0726 J/kg °C • D- 726 cal • E- none of themarrow_forward
- 1. A 250–g piece of gold is at 19 °C, 5192 J of energy is added to it by heat. The specific heat of gold is 129 J/(kg·°C). Calculate its final temperature. (A. 160.99 B. 179.99 C. 59.25) 2. How much heat is absorbed by 100 g of ice at -10⁰C to become water at 20⁰C?• Specific heat: Water (Ice) = 2,090 J/Kg•⁰C, Water (Liquid) = 4,186 J/Kg•⁰C• Latent heat of fusion (Water) = 3.33 x105 J/Kg( A. 4..38 X 10 ^ 4 B. 4.38 X 10 ^ 7 C. 4.76 X 10 ^ 5)arrow_forward• How does gas becomes ideal gas? • How does the work done in the following processes; a. Isobaric b. Isentropic c. Isothermal d. Isochronic e. Polytropicarrow_forwardTemperature• Define temperature.• Convert temperatures between the Celsius, Fahrenheit, and Kelvin scales.• Define thermal equilibrium.• State the zeroth law of thermodynamics.arrow_forward
- .•P60 Buckminsterfullerene, C60, is a large molecule consisting of 60 carbon atoms connected to form a hollow sphere. The diameter of a C60 molecule is about 7 x 10–10 m. It has been hypothesized that C60 molecules might be found in clouds of interstellar dust, which often contain interesting chemical compounds. The temperature of an interstellar dust cloud may be very low, around 3 K. Suppose you are planning to try to detect the presence of C60 in such a cold dust cloud by detecting photons emitted when molecules undergo transitions from one rotational energy state to another. Approximately, what is the highest-numbered rotational level from which you would expect to observe emissions? Rotational levels are l= 0,1,2,3,..arrow_forward© Example : • The base of an insect's leg is approximately spherical in shape ,with a radius of about 2x10-5 m. The 0.003 g mass of the insect is supported equally by the six legs .calculate the angle 0 for an insect on the surface of water .Assume the water temperature is 20°C ?arrow_forward3. For each of the situations below: • List which of the three methods of heat transfer are happening in that situation. Some situations will use more than one way of transferring heat. Which of the methods do you guess transfers the most heat in that situation? • For each method you find, describe how the heat transfer is happening. For example: if a hot cup is sitting on a cold desk, then heat is flowing via conduction from the hot cup to the cold desk. • If radiation is involved, explain what kind of light is being absorbed. (Remember, heat can be transferred by nonvisible light as well as visible light) a) A steaming pot of water is resting on a red-hot burner. b) A warm book is resting on the ground outside, in the Sun. c) You microwave a bowl of soup until it is very hot. You leave it resting on the counter to cool down. d) You sit alone in a cool room. e) On a hot summer day, you jump into a swimming pool. f) You roast a hot dog over a fire. g) Make up a situation of your…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
Thermal Expansion and Contraction of Solids, Liquids and Gases; Author: Knowledge Platform;https://www.youtube.com/watch?v=9UtfegG4DU8;License: Standard YouTube License, CC-BY