
The pV diagram in Figure 16.19 shows a
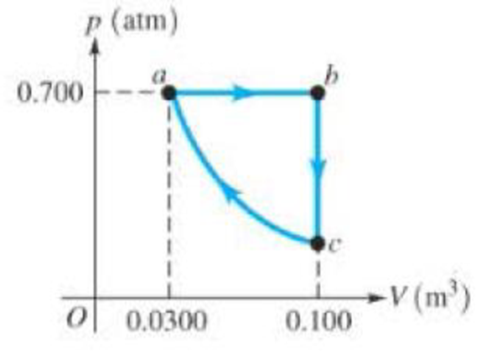
Figure 16.19
Problem 43.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
College Physics (10th Edition)
Additional Science Textbook Solutions
Physics for Scientists and Engineers with Modern Physics
Tutorials in Introductory Physics
Conceptual Integrated Science
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Physics (5th Edition)
University Physics with Modern Physics (14th Edition)
- A system consisting of 0.0816 moles of a diatomic ideal gas is taken from state A to state C along the path in the figure below. (b) What is the lowest temperature of the gas during this process? In kelvin. (c) Find the change in internal energy of the gas in going from A to C. Hint: Adapt the equation (for the change in internal energy of a monatomic ideal gas) ΔU=3/2nRΔT=3/2Δ(PV)=3/2(PcVc-PaVa) to a diatomic ideal gas. In joules. (d) Find the energy delivered to the gas in going from A to C. In joules.arrow_forwardAn ideal diatomic gas has an initial pressure of 1.00×10°Pa , an initial volume of 2.00m', and an initial temperature of 300K. (This is point 1 on the pV-diagram.) The gas has an isochoric increase in pressure to 2.00×10°P. . (This is point 2 on the pV-diagram.) The gas then has an isothermal expansion to a volume of 3.00m'. (This is point 3 on the pV-diagram.) The pressure is then reduced adiabatically back down to its original pressure of 1.00×10°P.. (This is point 4 on the pV-diagram.) Finally, the gas has an isobaric decrease in volume to its original volume of 2.00m. (The gas is back to point 1 on the pV-diagram.) а. Fill in the missing values on the following table. Point Volume, Pressure, Temperature, v (m²) p(10ʻPA) T(K) 1 2.00 1.00 300 2 2.00 3 3.00 4 1.00 b. Fill in the values for each of the processes in the following table. (These values correspond to the First Law of Thermodynamics written as: AE, =W +Q.) Process Change in internal Work done to gas, Heat added to gas,…arrow_forwardA heat engine takes 0.350 molmol of a diatomic ideal gas around the cycle shown in the pV-diagram of (Figure 1). Process 1→21→2 is at constant volume, process 2→32→3 is adiabatic, and process 3→13→1 is at a constant pressure of 1.00 atmatm. The value of γγ for this gas is 1.40. Figure 1 of 1 Part A Find the pressure at points 1, 2, and 3. Finally, I AM BLIND!! I can't read the math equations you have on here they are all formatted in pictures. is there a way you could just write it all out in text like I am currently doing with this post? Thanksarrow_forward
- The pV diagram in (Figure 1) shows a process abc involving 0.550 mol of an ideal gas. For related problem-solving tips and strategies, you may want to view a Video Tutor Solution of A cyclic process. Figure 8.0 6.0 4.0 p(Pax 105) 2.0 O a 0.020 0.040 0.060 b 0.080 1 of 1 V(m³) ▶ Part A What was the temperature of this gas at points a, b, and c? Enter your answers separated by commas. Ta, Tb, Tc= 535,9355,14968 V Submit Previous Answers Request Answer Part B X Incorrect; Try Again; 5 attempts remaining Part C ΑΣΦ ΑΣΦ Q = 3.5104 How much heat had to be put in during the process to increase the internal energy of the gas by 1.00×104 J ? Express your answer in joules. Provide Feedback Submit Previous Answers Request Answer 國」? ? A p V diagram of a two-step process. X Incorrect; Try Again; 4 attempts remaining K Next >arrow_forwardA gas expands from I to F in the figure below. The energy added to the gas by heat is 422 J when the gas goes from I to F along the diagonal path. Three paths are plotted on a PV diagram, which has a horizontal axis labeled V (liters), and a vertical axis labeled P (atm). The green path starts at point I (2,4), extends vertically down to point B (2,1), then extends horizontally to point F (4,1). The blue path starts at point I (2,4), and extends down and to the right to end at point F (4,1). The orange path starts at point I (2,4), extends horizontally to the right to point A (4,4), then extends vertically down to end at point F (4,1). (a) What is the change in internal energy of the gas? J(b) How much energy must be added to the gas by heat for the indirect path IAF to give the same change in internal energy? Jarrow_forwardConsider the thermodynamic process, A->B->C->A shown above. The heat absorbed during A->B is 591J. If the change in internal energy during B->C is 4146J, What is the change in internal energy in SI units during C->A? Express only the number of your answer with 4 significant figures.arrow_forward
- A monatomic ideal gas undergoes an isothermal expansion at 300 K, as the volume increased from 0.020 to [2*v1] m³.The final pressure is 120.61kPa. The ideal gas constant is R = 8.314 J/mol · K. %3D What is the heat transfer Q to the gas in kilojoules? Please enter your numerical answer with two decimal places. (for example for 2090 J, enter 2.09 )arrow_forwardProblem 11: A diatomic ideal gas goes through the cycle a → b → c → d → a as shown in the figure. Processes ab and cd are isothermal and occur at temperatures TH = 390 K and TC = 288K, respectively. There are n = 45 moles of this gas in the system, and the initial volume is Va = 0.079 m3. Calculate the total work W done in the entire cycle, in joules.arrow_forwardA quantity of a monatomic ideal gas undergoes a process in which both its pressure and volume are increased by a factor of n = 9 as shown in the figure below. What is the energy absorbed by heat into the gas during this process? Hint: The internal energy of a monatomic ideal gas at pressure P and occupying volume V is given by U = 3PV. 2 (Give your answer as a multiple of Povo.) x Your response differs significantly from the correct answer. Rework your solution from the beginning and check each step carefully. x PoVo P nPo 8 Po Vo nVo Varrow_forward
- A gas sample undergoes the cyclic process ABCA shown in the figure (Figure 1), where AB lies on an adiabat (a specific-heat ratio of y = 1.4). The pressure at point A is 56 kPa. Figure Pressure, p 0 B 2 3 4 Volume, V (L) A 5 1 of 1 Part A Find the pressure at B. Express your answer in kilopascals. PB 530 kPa Submit Part B W Correct Find the net work done on the gas. Express your answer in joules. = Previous Answers Submit VE ΑΣΦ Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining Return to Assignment Provide Feedback ? Jarrow_forwardA container is filled with an ideal diatomic gas to a pressure and volume of P₁ and V₁, respectively. The gas is then warmed in a two-step process that increases the pressure by a factor of four and the volume by a factor of five. Determine the amount of energy transferred to the gas by heat if the first step is carried out at constant volume and the second step at constant pressure. (Use any variable or symbol stated above as necessary.) Q =arrow_forward2 moles of a monatomic ideal gas undergoes a cyclic process as depicted in the figure below. The processes AB and CD are isobaric and the process DA is adiabatic. For the given values PA= 11.5 atm, VA= 5.7 L, V3= 2.85 L, Pc=34.5 atm, and Vc=1.476 L answer the following questions. (use R=8.314 J 1 atm = 1.013x105 Pa, 1 L = 10-3 m³) . mol · K' Volume 1. Calculate the temperature TA= K 2. What type of process is the process BC? 3. Calculate the work done by the gas in the process DA. WDA = 4. Calculate the magnitude of the net heat entering the cycle. Q = 5. Calculate the magnitude of the net heat leaving the cycle. Qc = 6. Calculate the net work done by the gas. W= 7. Calculate the thermal efficiency of the cycle. e = % 8. Calculate the change in the entropy in the process AB. Include the sign (positive or negative) in your answer as well. ASAB = Karrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





