
Concept explainers
Predict the major products of the following reactions:
(a)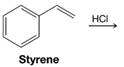
(b) 2-Bromo-1-phenylpropane
(c)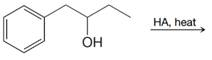
(d) Product of (c) + HBr
(e) Product of (c) +
(f) Product of (c) +
(g) Product of (f)
(h)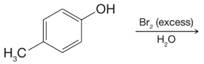
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Fundamentals of Heat and Mass Transfer
Chemistry: Matter and Change
Chemistry: The Central Science (14th Edition)
Principles of Chemistry: A Molecular Approach (3rd Edition)
General, Organic, and Biological Chemistry (3rd Edition)
General Chemistry: Atoms First
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forward2) Which of the following compounds is the most acidic? A D B C 3) Which of the following compounds will undergo an SN1 process most readily? CI A CI B CI C D CI D 4) Which of the following compounds will react most rapidly with ethyl chloride in the presence of aluminum trichloride? A CN B Me C OMe D CIarrow_forwardThe MAJOR product of the following reaction is: (a) HO (b) a b n O P CH3 OH H3C S S S CH3 CH3 CH3 CH3 +R, S H3O+ CH3 (c) (d) H3C H3C H3C CH3 R S OH CH3 OH +R +S CH3arrow_forward
- What is the product of the following reaction? H30*,H2O OH (а) НО° HO (b) (d) (c) HO HOarrow_forwardNaH HO HO PBR3 A (a) Name the functional groups in compounds A and B, including, if relevant, whether they are primary (1°), secondary (2°) or tertiary (3'). (b) Propose a reagent that could be used to convert A into B. (c) Draw the structure of the intermediate C formed by reaction of compound A with NaH. (d) Propose the structure of the organic compound D formed by reaction with PB.3. (e) Could HBr be used for the conversion of A into D? Explain very briefly.arrow_forwardWhat is the major product from the following reaction? 1) 2) H202, NaOH OH OH OH (A) (B) (D) O A В O C O D 1 ptsarrow_forward
- Determining the Regioselectivity of Opening an Epoxide Ring What product is formed when 2,2-dimethyloxirane is treated with each set of reagents: −OCH3 followed by H2O, or CH3OH and H2SO4?arrow_forwardWhich of the following compounds is generated by the reaction below? он (1) B2H6. diglyme но (2) H2O2, NAOH/H2O OH (A) (B) (C) (D) OCompound C Compounds A and C O Compound B OCompounds A and Darrow_forwardTreatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products (M and N) with molecular formula C6H12. The 1H NMR spectra of M and N are given below. Propose structures for M and N and draw a mechanism to explain their formation.arrow_forward
- Following is the structural formula of the principal sex pheromone of the Douglas fir tussock moth (Orgyia pseudotsugata), a severe defoliant of the fir trees of western North America. (Z)-6-Heneicosene-11-one Several syntheses of this compound have been reported, starting materials for three of which are given here. (a) `H + OH Br (b) HO Br (c) Br + Show a series of steps by which each set of starting materials could be converted into the target molecule.arrow_forwardHow would you carry out the following reactions? More than one step may be required. (a) 3-Hexyne →→ 3-Hexanone (b) Benzene →→ m-Bromoacetophenone (c) Bromobenzene →→ Acetophenone (d) 1-Methylcyclohexene →→ 2-Methylcyclohexanonearrow_forwardWhen A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With this information, write the correct structure of B and propose the reaction mechanism (step by step, with the correct use of arrows) to obtain B.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

