
Concept explainers
PRACTICE PROBLEM 15.15
Suppose you needed to synthesize m-chloroethylbenzene from benzene.
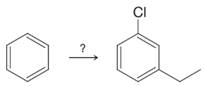
You could begin by chlorinating benzene and then follow with a Friedel–Crafts alkylation using chloroethane and
(a) Why will neither method give the desired product?
(b) There is a three-step method that will work if the steps are done in the right order. What is this method?
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry
Additional Science Textbook Solutions
General Chemistry: Atoms First
Chemistry: The Central Science (13th Edition)
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry For Changing Times (14th Edition)
Introductory Chemistry (5th Edition) (Standalone Book)
- Show how you would synthesize the following compounds, starting with benzene or toluene and any necessary acyclic reagents. Assume para is the major product (and separable from ortho) in ortho, para mixtures.(a) ethoxybenzene (b) 1,2-dichloro-4-nitrobenzene (c) 1-phenylpropan-2-olarrow_forwardShow how you would synthesize the following compounds, starting with benzene or toluene and any necessary acyclic reagents. Assume para is the major product (and separable from ortho) in ortho, para mixtures.(a) 1-phenyl-1-bromobutane (b) 1-phenyl-1-methoxybutane (c) 3-phenylpropan-1-olarrow_forwardNucleophilic addition to ketone - Match the reagent with the reaction. (each used once) OH H3CO OCH3 OH HO. OH (A) (B) PPh3=CHCH3 1) CH3MgBr 2) H3O+ (C) CH3-OH (D) NaBH4 (E) 1) NaCN (aq) 2) H3O+ (F) H₂O OH CO₂Harrow_forward
- Practice Problem 19.54 Z Your answer is partially correct. Try again. Predict the major product(s) (A - K) from the treatment of acetone with the following compounds (a-c): NH2 HO Eto OEt A: B: C: D: E: F: OH OH но CN G: H: I: J: (a) [H*], excess EtOH, (-H20) Major Product(s): (ь) NaBH4, Meон B Major Product(s): (c) LAH followed by H20 Major Product(s): SHOW HINTarrow_forwardA key step in the synthesis of naproxen, an NSAID more commonly known by its brand name, Aleve (Section 3.9), is a coupling reaction of 2-bromo-6-methoxynaphthalene to form 2-methoxy-6-vinylnaphthalene. Show three different coupling reactions, and the required reagents, that could be used tocarry out this step.arrow_forwardIn an aqueous solution containing sodium bicarbonate, aniline reacts quickly withbromine to give 2,4,6-tribromoaniline. Nitration of aniline requires very strong conditions,however, and the yields (mostly m-nitroaniline) are poor.(a) What conditions are used for nitration, and what form of aniline is present under theseconditions?arrow_forward
- Illustrate the resonance effect of the methoxy group -OCH3, on the structure of the benzene ring. Draw all the oissuvke resonance forms of methoxybenzene, including the hybrid Based on the structures, explain how the presence of the -OCH3 group affects: (i) the reactivity of the benzene ring towards electrophilic attack (ii) the orientation or point of attack of an incoming electrophilic reagent on the benzene ring.arrow_forward8. (a) Benzene derivatives exhibit medium to strong absorption in UV-region. Explain why aniline and phenoxide ion have strong UV-absorptions.arrow_forward18.26 Predict the products of the following ether cleavage reactions: LOCH CH3 OC(CH3)3 (a) HI CF3CO,H, ? (b) H2O HI HI (c) Н,С— СНОСH,CH, (d) (CH3);CCH,OCH,CH3 H20 H2Oarrow_forward
- Propose the reagents in best order for the synthesis of the following compound from benzene. CI A) B) 1) CH3CH2Cl, AlCl3 2) Cl₂, AlCl3 3) CH3COCI, AlCl3 1) CH3CH2Cl, AlCl3 2) CH3COCI, AlCl3 3) Cl₂, AlCl3 D) 1) CH3CH2Cl, AlCl3 2) Fuming H₂SO4 2) Cl₂, AlCl3 3) CH3COCI, AlCl3 4) Dilute H2SO4 E) C) 1) CH3COCI, AlCl3 2) Zn(Hg), HCl, Heat 3) Fuming H2SO4 4) Cl₂, AlCl3 5) Dilute H2SO4 1) CH3COCI, AlCl3 2) Cl₂, AlCl3 3) CH3CH2Cl, AlCl3 4) Zn(Hg), HCl, Heatarrow_forwardHow are following obtained: (i) Toluene from phenol (ii) Phenol from Aniline.arrow_forwardNucleophilic aromatic substitution provides one of the common methods for making phenols. ) Show how you would synthesize the following phenols, using benzene or toluene as your aromatic starting material, and explain why mixtures of products would be obtained in some cases. (a) m-cresol (b) p-n-butylphenolarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





