
Concept explainers
Interpretation:
Difference that can exist between Arrhenius and Bronsted-Lowry definitions of an acid has to be written.
Concept Introduction:
Arrhenius gave broader picture and suggested that acids are able to release excess of
Bronsted-Lowry acid outlined the definition of acids that donate
Explanation of Solution
In accordance with Bronsted definition, the most usual type of acid-base reaction involves lone pair of base that reaches out for an acidic proton. Once deprotonation has occurred species assumes a negative charge and is referred to as the conjugate base of acid and other species with a positive charge as a result of proton acceptance is termed conjugate acid of given base. For example;
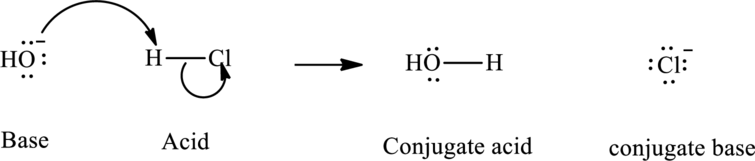
In Arrhenius concept, the acid species is one that can make water to behave as Bronsted base so that it accepts the proton and forms
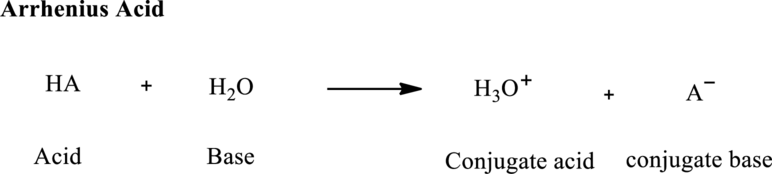
Thus all
Want to see more full solutions like this?
Chapter 15 Solutions
EBK FOUNDATIONS OF COLLEGE CHEMISTRY
- A sample of vinegar has a hydronium-ion concentration of 3.0 103 M. What is the pH of the vinegar?arrow_forwardExplain the difference between a strong acid and a weak acid.arrow_forward. In each of the following chemical equations, identify the conjugate acid-base pairs. a. NH3(aq)+H2O(l)NH4+(aq)+OH(aq) b. PO43(aq)+H2O(1)HPO42(aq)+OH(aq) c. C2H3O2(aq)+H2O(l)HC2H3O2(aq)+OH(aq)arrow_forward
- Which of the following substances are acids in terms of the Arrhenius concept? Which are bases? Show the acid or base character by using chemical equations. a P4O10 b Na2O c N2H4 d H2Tearrow_forward8-13 Define (a) an Arrhenius acid and (b) an Arrhenius base.arrow_forwardWhat is meant by “pH”? True or false: A strong acid always has a lower pH than a weak acid does. Explain.arrow_forward
- Define each of the following: a. Arrhenius acid b. BronstedLowry acid c. Lewis acid Which of the definitions is most general? Write reactions to justify your answer.arrow_forwardEthanol (ethyl alcohol), CH3CH2OH, can act as a BrnstedLowry acid. Write the chemical equation for the reaction of ethanol as an acid with hydroxide ion, OH. Ethanol can also react as a BrnstedLowry base. Write the chemical equation for the reaction of ethanol as a base with hydronium ion, H3O+. Explain how you arrived at these chemical equations. Both of these reactions can also be considered Lewis acid base reactions. Explain this.arrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





