
Concept explainers
Interpretation:
The solubility of potassium bromide (KBr) and potassium nitrate (KNO3) at 80°C needs to be described.
Concept introduction:
Solubility: Solubility can be expressed as maximum amount of solute that can dissolve in known amount of solvent at a given temperature and pressure.
Effect of temperature on solubility: Solubility is affected by increasing the temperature of the solvent as the kinetic energy of its particles is increased, thus more-frequent collisions occur. This is why many substances are more soluble at high temperatures.
Answer to Problem 91A
From the graph:
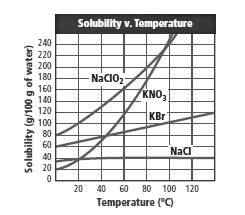
It can be seen that the solubility of potassium nitrate is nearly double to the solubility of potassium bromide at 80°C.
Explanation of Solution
From the graph,
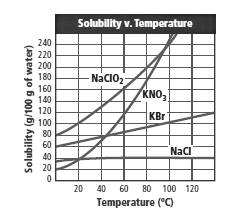
The values of the solubility at different temperature can be obtained. At 80°C, the solubility of KBr is almost 95 g in 100 g H2O whereas the solubility of KNO3 is nearly 170 g in 100 g H2O which is nearly twice as high as the solubility of KBr.
Chapter 14 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Organic Chemistry
Inorganic Chemistry
General, Organic, and Biological Chemistry (3rd Edition)
Organic Chemistry (9th Edition)
Chemistry: The Central Science (13th Edition)
Chemistry: The Central Science (14th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





