
Concept explainers
(a)
Interpretation:
The following alcohol should be classified as primary, secondary and tertiary alcohol:
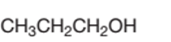
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds are said to be
Answer to Problem 14.29P
Propanol is a primary alcohol.
Explanation of Solution
Alcohols are the organic compounds with the general formula R-OH. Here, the −OH group (hydroxyl group) is directly bonded to the carbon atom of the parent chain. On the basis of the carbon atom to which the −OH group is bonded, alcohols can be classified as primary, secondary and tertiary alcohols.
- Primary alcohol = The −OH group must be bonded to the primary carbon atom (bonded with one other C atom).
- Secondary alcohol = The −OH group must be bonded to the secondary carbon atom (bonded with two other C atoms).
- Tertiary alcohol = The −OH group must be bonded to a tertiary carbon atom (bonded with three other C atoms).
In the given alcohol, the −OH group is bonded to a primary C atom which is bonded with one other C atom hence, it should be primary alcohol.
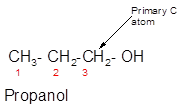
(b)
Interpretation:
The following alcohol should be classified as primary, secondary and tertiary alcohol:
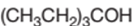
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds are said to be organic chemistry. The molecular formula of the organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule.
Answer to Problem 14.29P
3-ethyl-3-pentanol is a tertiary alcohol.
Explanation of Solution
Alcohols are the organic compounds with the general formula R-OH. Here the −OH group (hydroxyl group) is directly bonded to the carbon atom of the parent chain. On the basis of the carbon atom to which the −OH group is bonded, alcohols can be classified as primary, secondary and tertiary alcohols.
- Primary alcohol = The −OH group must be bonded to the primary carbon atom (bonded with one other C atom).
- Secondary alcohol = The −OH group must be bonded to the secondary carbon atom (bonded with two other C atoms).
- Tertiary alcohol = The −OH group must be bonded to a tertiary carbon atom (bonded with three other C atoms).
In the given alcohol, the −OH group is bonded to a tertiary C atom which is bonded with 3 other C atoms hence, it should be tertiary alcohol.

(c)
Interpretation:
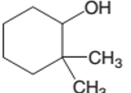
The following alcohol should be classified as primary, secondary and tertiary alcohol:
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds are said to be organic chemistry. The molecular formula of the organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule.
Answer to Problem 14.29P
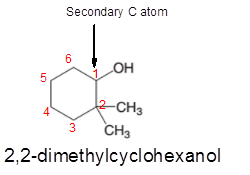
2,2-dimethylcyclohexanol is a secondary alcohol.
Explanation of Solution
Alcohols are the organic compounds with the general formula R-OH. Here the −OH group (hydroxyl group) is directly bonded to the carbon atom of the parent chain. On the basis of the carbon atom to which the −OH group is bonded, alcohols can be classified as primary, secondary and tertiary alcohols.
- Primary alcohol = The −OH group must be bonded to the primary carbon atom (bonded with one other C atom).
- Secondary alcohol = The −OH group must be bonded to the secondary carbon atom (bonded with two other C atoms).
- Tertiary alcohol = The −OH group must be bonded to a tertiary carbon atom (bonded with three other C atoms).
In the given alcohol, the −OH group is bonded to a secondary C atom which is bonded with two other C atoms hence, it should be secondary alcohol.
(d)
Interpretation:
The following alcohol should be classified as primary, secondary and tertiary alcohol:
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds are said to be organic chemistry. The molecular formula of the organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule.
Answer to Problem 14.29P
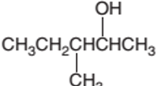
3-methyl-2-pentanol is a secondary alcohol.
Explanation of Solution
Alcohols are the organic compounds with the general formula R-OH. Here the −OH group (hydroxyl group) is directly bonded to the carbon atom of the parent chain. On the basis of the carbon atom to which the −OH group is bonded, alcohols can be classified as primary, secondary and tertiary alcohols.
- Primary alcohol = The −OH group must be bonded to the primary carbon atom (bonded with one other C atom).
- Secondary alcohol = The −OH group must be bonded to the secondary carbon atom (bonded with two other C atoms).
- Tertiary alcohol = The −OH group must be bonded to a tertiary carbon atom (bonded with three other C atoms).
In the given alcohol, the −OH group bonds to a secondary C atom which is bonded with two other C atoms hence it should be secondary alcohol.
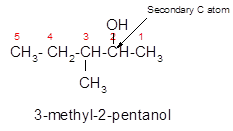
Want to see more full solutions like this?
Chapter 14 Solutions
General, Organic, & Biological Chemistry
- Which is NOT a physical property of alcohols or phenols? O Phenols are generally only slightly soluble in water. O The hydroxyl group of an alcohol is nonpolar. The solubilities of normal primary alcohols in water decrease with increasing molecular weight. Boiling points of normal primary alcohols increase with increasing molecular weight.arrow_forwardPredict which member of each group is most soluble in water, and explain the reasons for your predictions.chlorocyclohexane, cyclohexanol, or cyclohexane-1,2-diolarrow_forwardClassify these alcohols as primary (1°), secondary (2°), or tertiary (3°). 1° 2° 3° CH,CH, CH,COH ČH,CH, OH CH,CHCHCH, CH, CH,CH, CH,CCH,OHarrow_forward
- Which of these compounds is a properly named 2° alcohol? a) 1,3-Propandiol HO HO b) 2-Methyl-3-methyl-3-pentanol CH3 H3C CH3 OH c) 1-Propanolarrow_forwardDifferentiate the three types of alcohols. Give an example of eacharrow_forwardWhich of the following alcohols is the MOST soluble in water? O 2-methyl-2-pentanol O 4-methyl-2-pentanol O 3-methyl-1-butanol O 2-methyl cyclohexanolarrow_forward
- Select the correct IUPAC name for the branched alcohol. OH HC OH CH CH CH CH CH HC CH CH The correct IUPAC name is: A) 3,4,6-trimethyl-4-propyl-2-heptanol B) 3,4,6-trimethyl-4-propyl-3-heptanol C) 2,4,5-trimethyl-4-propyl-5-heptanol D) 2-ethyl-3,5-dimethyl-3-propyl-2-hexanolarrow_forwardDefine the Properties of Alcohols ?arrow_forwardName each of the following alcohols. HO CHI CHO он OH H2d-CH-CH3 НЗС H3C-CH2-CH2-CH-CH2-CH2-OH H2C-CH3 왜 I H3C-CH2-C-CH2-CH3 CH3arrow_forward
- 3. Complete the following intramolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) Cy-C-CH-oH 18UC b) CHy CH-Cy-CHy 180C OH c) 180c 4. Complete the following intermolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy-CH-OH b) Ho SわふわSわ 5. Complete the following oxidation reactions for alcohols. Draw the structure of the product. Name the reactant and identify the type of compound formed in the product. a) hparrow_forward1. Br + CH3O°Na* methanol 2. CI NH3 ethanol 3. CI CH3 O'Na* ethanolarrow_forwardWhat are the best reagents to make the following product? NO O acetyl chloride and AIC13 O SO3 and H2SO4 O 1-chloropropanc and AICI3 O HNO3 and H2S04arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





