
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 48P
Give reagents suitable for carrying out each of the following reactions, and write the major
organic products. If an ortho, para mixture is expected, show both. If the meta isomer is the
expected major product, write only that isomer.
Nitration of nitrobenzene
Bromination of toluene
Bromination of (trifluoromethyl)benzene
Sulfonation of anisole
Sulfonation of acetanilide (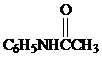 )
)
Chlorination of bromobenzene
Friedel–Crafts alkylation of anisole with benzyl chloride
Friedel–Crafts acylation of benzene with benzoyl chloride (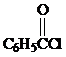 )
)
Nitration of the product from part (h)
Clemmensen reduction of the product from part (h)
Wolff–Kishner reduction of the product from part (h)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Outline a synthesis of each of the following compounds from isopropyl alcohol. A compound prepared in one part can be used as a reactant in another. (Hint: which of the compounds shown can serve as a starting material to all others?)
Provide the structure of the major organic product of the following reaction and? explain the stereochemistry which results in this product. 2-Pentanol reacting with 1.) PBr3, pyridine 2.) NaCN
the following reaction scheme leads to the formation of compound B. give the structure of the final products and of the intermediate product A and justify, using the mechanism, the formation of these
Chapter 13 Solutions
Organic Chemistry - Standalone book
Ch. 13.2 - Based on Hammonds postulate which holds that the...Ch. 13.3 - Prob. 2PCh. 13.3 - Using : O =N+= O : as the electrophile, write a...Ch. 13.4 - Prob. 4PCh. 13.5 - Prob. 5PCh. 13.6 - Prob. 6PCh. 13.6 - Write a reasonable mechanism for the formation of...Ch. 13.6 - tert-Butylbenzene can be prepared by alkylation of...Ch. 13.6 - Prob. 9PCh. 13.7 - The reaction shown gives a single product in 88...
Ch. 13.7 - Prob. 11PCh. 13.8 - Using benzene and any necessary organic or...Ch. 13.10 - Prob. 13PCh. 13.11 - Prob. 14PCh. 13.12 - Prob. 15PCh. 13.12 - Prob. 16PCh. 13.13 - Prob. 17PCh. 13.13 - Prob. 18PCh. 13.14 - Reaction of chlorobenzene with p-chlorobenzyl...Ch. 13.15 - Prob. 20PCh. 13.15 - Prob. 21PCh. 13.15 - Prob. 22PCh. 13.16 - Prob. 23PCh. 13.16 - Prob. 24PCh. 13.17 - Prob. 25PCh. 13.18 - Prob. 26PCh. 13.19 - Write the structure of the expected product from...Ch. 13.20 - Prob. 28PCh. 13.20 - Prob. 29PCh. 13.21 - Prob. 30PCh. 13.21 - Offer an explanation for the observation that...Ch. 13.21 - Prob. 32PCh. 13 - Write the structure of the organic product in each...Ch. 13 - Prob. 34PCh. 13 - Prob. 35PCh. 13 - Prob. 36PCh. 13 - Prob. 37PCh. 13 - Prob. 38PCh. 13 - Prob. 39PCh. 13 - Treatment of the alcohol shown with sulphuric acid...Ch. 13 - Prob. 41PCh. 13 - Prob. 42PCh. 13 - Prob. 43PCh. 13 - Arrange the following five compounds in order of...Ch. 13 - Prob. 45PCh. 13 - Prob. 46PCh. 13 - Prob. 47PCh. 13 - Give reagents suitable for carrying out each of...Ch. 13 - Prob. 49PCh. 13 - Prob. 50PCh. 13 - Which is the best synthesis of the compound shown?Ch. 13 - What combination of acyl chloride or acid...Ch. 13 - A standard synthetic sequence for building a...Ch. 13 - Prob. 54PCh. 13 - Prob. 55PCh. 13 - Prob. 56PCh. 13 - Prob. 57PCh. 13 - Prob. 58PCh. 13 - Prob. 59PCh. 13 - Prob. 60DSPCh. 13 - Prob. 61DSPCh. 13 - Prob. 62DSPCh. 13 - Prob. 63DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Compounds with more than one hydroxyl group can react with thionyl chloride of differently from simple alcohols. In reaction with thionyl chloride, butane-1,2-diol gave a single organic product with 85% yield, according to the following balanced equation: Suggest through a mechanism, a reasonable structure for this productarrow_forwardProvide all organic products for the following reactionarrow_forwardplease help with the following Ochem question... In theory a poorly planned Claisen condensation between 2 different esters, isopropyl butanoate and tertbutyl propanoate, can produce how many different products? Provide the bond line structures and the IUPAC names of the products obtainedarrow_forward
- Suggest reasonable mechanisms for each of the following reaction.arrow_forwardQuestion (a) Discuss why the reactivity of nitration towards benzene increases when a mixture concentrated nitric acid (HNO:) mixed with sulphuric acid (H2SO4). You may explain it from the aspect of electrophile formed in the reaction between HNO3 and H2SO4.arrow_forwardHeterocyclic compounds are present in naturally occurring substances and their syntheticcompounds are produced for various industrial applications. Furan is one such heterocycliccompound which is more reactive than benzene towards electrophilic aromatic substitutionreaction.1.Analyze the different substitution products formed in case of furan when it is reacted withiodine. Write the reaction mechanism involved when furan reacted with iodine andevaluate the formation of products.arrow_forward
- Give the major organic product generated by each of the following reactionsarrow_forwardGive the structure of the product and/or intermediates of the following reactions. Indicate, whenappropriate, both regiochemistry and stereochemistryarrow_forward(A) Provide the major organic product for the reaction below (B) Would the product be optically active of optically in active?arrow_forward
- (a) What reagents would be used for the conversion of alkene A into the target? (b) What reaction is involved in the conversion of alcohol B into alkene A? Suggest a reagent that might affect this transformation. (c) Give a retrosynthetic analysis showing the disconnection of B, the synthons produced that lead to the synthetic equivalents given (draw their structures).arrow_forwardPlease don't provide handwritten solution .... What is the major organic product from the following reaction?arrow_forwardGive the clear handwritten answer (provide full reaction mechanism for the following reaction)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License