
Chemistry
4th Edition
ISBN: 9780078021527
Author: Julia Burdge
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.2, Problem 1PPB
Practice Problem BUILD
BUILD
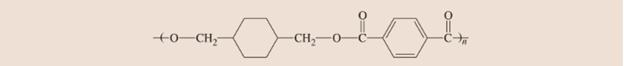
Kodel is a polymer used to make stain-resistant carpets. Draw the structure of the monomer. Is this an addition polymer or a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the properties below apply to the following polymer?
farm
Choose all that apply.
a polyester
an addition polymer
a polyamide
a substituted polyethylene
a polymer formed by splitting out water
a which of the following monomers could form an addition polymer?
CH4
O HO-CH(CH3)-COOH
O HỌ-CH,CH, CH3
O C₂F4
Submit
b which of the following monomers could form a condensation polymer?
CH4
HO-CH(CH3)-COOH
O HỌ-CH2-CH, CH3
O C₂F4
Which of the properties below apply to the following polymer?
fomm
Choose all that apply.
O a polyester
O an addition polymer
a polymer formed by splitting out water
O a substituted polyethylene
O a polyamide
Chapter 12 Solutions
Chemistry
Ch. 12.1 - Practice ProblemATTEMPT Saran Wrap, the original...Ch. 12.1 - Prob. 1PPBCh. 12.1 - Practice ProblemCONCEPTUALIZE Which diagram best...Ch. 12.1 - Prob. 1CPCh. 12.1 - Prob. 2CPCh. 12.2 - Prob. 1PPACh. 12.2 - Practice ProblemBUILD Kodel is a polymer used to...Ch. 12.2 - Prob. 1PPCCh. 12.3 - Prob. 1PPACh. 12.3 - Prob. 1PPB
Ch. 12.3 - Prob. 1PPCCh. 12.3 - Prob. 1CPCh. 12.3 - Prob. 2CPCh. 12.4 - Prob. 1PPACh. 12.4 - Prob. 1PPBCh. 12.4 - Prob. 1PPCCh. 12.5 - Prob. 1PPACh. 12.5 - Prob. 1PPBCh. 12.5 - Prob. 1PPCCh. 12.6 - Prob. 1CPCh. 12.6 - Prob. 2CPCh. 12 - 12.1 Bakelite, the first commercially produced...Ch. 12 - Prob. 2QPCh. 12 - Prob. 3QPCh. 12 - Prob. 4QPCh. 12 - Prob. 5QPCh. 12 - Prob. 6QPCh. 12 - Prob. 7QPCh. 12 - Prob. 8QPCh. 12 - Prob. 9QPCh. 12 - Prob. 10QPCh. 12 - Bakelite. described in Review Question 12.1, is...Ch. 12 - Is a normal liquid isotropic or anisotropic? How...Ch. 12 - Prob. 13QPCh. 12 - 12.14 Would each of these molecules be likely to...Ch. 12 - Prob. 15QPCh. 12 - 12.16 Would an ionic compound form a liquid...Ch. 12 - Prob. 17QPCh. 12 - Prob. 18QPCh. 12 - Prob. 19QPCh. 12 - 12.20 What are some advantages and disadvantages...Ch. 12 - Prob. 21QPCh. 12 - 12.22 How does an measure the peak and valley...Ch. 12 - Name four allotropic forms of carbon.Ch. 12 - Prob. 24QPCh. 12 - Prob. 25QPCh. 12 - Prob. 26QPCh. 12 - 12.27 What type of intermolecular forces holds the...Ch. 12 - Prob. 28QPCh. 12 - Prob. 29QPCh. 12 - Prob. 30QPCh. 12 - Prob. 31QPCh. 12 - Prob. 32QPCh. 12 - Prob. 33QPCh. 12 - Prob. 34QPCh. 12 - Prob. 35QPCh. 12 - Prob. 36QPCh. 12 - What types of bonding (covalent, ionic, network,...Ch. 12 - Draw representations of isotactic, syndiotactic....Ch. 12 - Prob. 39QPCh. 12 - Draw representations of block copolymers and graft...Ch. 12 - Prob. 41APCh. 12 - 12.42 Would the compound shown form a liquid...Ch. 12 - Prob. 43APCh. 12 - Prob. 44APCh. 12 - Fluoride ion is commonly used in drinking water...Ch. 12 - Prob. 1SEPPCh. 12 - Prob. 2SEPPCh. 12 - Prob. 3SEPPCh. 12 - Prob. 4SEPP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of these, if any, is different? A) B) CI CI CI C) D) Polymer of Polymer of CI E) All the above are the samearrow_forwardD Listen Which of the following statements about branched polymers is TRUE? Materials made from branched polymers are less pliable than those made from linear polymers Most thermoplastics are branched polymers HDPE is an example of a branched polymer Branched polymers tend to have lower densities than linear polymers Plastics made from branched polymers can be reshaped De VSEPR Shapesarrow_forwardWhat monomer or monomers were used to create this polymer? correct monomer(s) not shown andarrow_forward
- Which polymer uses this monomer? HO HO A) B) 3 C) D)arrow_forwardWhich of the properties below apply to the following polymer? -(CH) -(CH) Choose all that apply. a polymer formed by splitting out water an addition polymer a polyester a substituted polyethylene a polyamidearrow_forwardSelect the single best answer. Choose the definition that best describes a polymer: a acidic compounds made up of repeating monomers b very large molecules made up of repeating units c a molecule containing oxygen and carbon only d synthetic molecules with a large molar massarrow_forward
- QUESTION 5 Below is the polymer Teflon (used for non-stick coating) (H) F F F The monomer used to make the polymer Teflon is CH2=CHF U CH2=CHCN CH2=CF2 O CF2=CF2arrow_forwardThere are two (2) types of polymerization; addition or linear and cross-linked or condensation. Addition polymers are thermoplastics and cross-linked polymers are thermosetting materials. True Falsearrow_forwardPolymer Foams also known as foamed polymers of engineering which has wide variety of consumer products. What are the TWO (2) examples of polymer member from polymer foam class? Natural rubber and silicon rubber Epoxies and polycarbonate Polyethylene and polypropylene Polyester and polystyrenearrow_forward
- Which statement is true about addition polymerization but not condensation polymerization? 1.) only one type of monomer is used 2.) many monomers link together to form polymers3.) only one product is formedarrow_forwardNylon is a polymer made up of repeating______units linked via_____bonds 1) phosphorus; oxide 2) oxygen; amine 3) benzene; amide 4) carbon; amidearrow_forwardMolecule X is a linear polymer that is the main component of wood and cotton. What is the common (not IUPAC) name of X? Note: be as specific as you can be. A name that is too general, like "big molecule" or "protein" will be marked wrong. Select the sketch that shows what X looks like when added to water. (Choose one) i i Each blue shape stands for a monomer. Suppose a molecule of X is completely hydrolyzed, for example by an enzyme, or by acid and heat. List the common (not IUPAC) names of the molecules that will be released. 0 0arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY