
Concept explainers
Interpretation:
The term resonance is to be described giving examples.
Concept introduction:
The concept resonance comes into play when properties of a molecule can’t be explained by a single structure.
Answer to Problem 53A
Resonance is a phenomenon of having more than one structure of a molecule. The various structures are resonating structures and the most stable structure is resonance hybrid. The molecules or ions which show resonance are: benzene, carbonate ion, sulphate ion, nitrate ion.
Explanation of Solution
Resonating structures of sulphate ion:
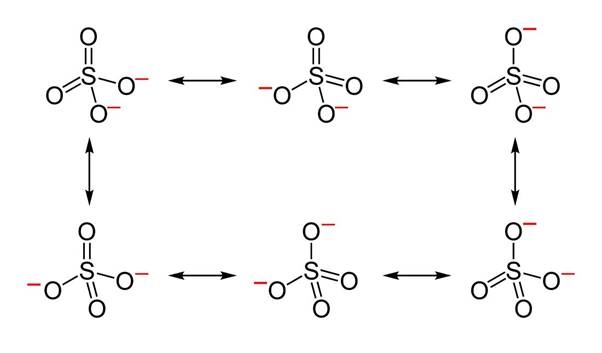
Sulphur has six valence electrons. One electron is shared by each oxide ion and two electrons are shared by oxygen atom and no electron pairs are left unshared.
Resonating structures of phosphate ion:
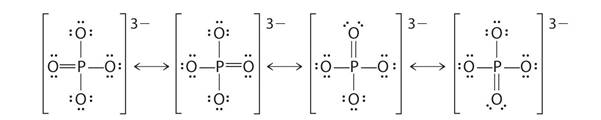
Phosphorus has five valence electrons. One electron is shared by each oxide ion and two electrons are shared by oxygen atom and no electron pairs are left unshared.
Resonating structures of
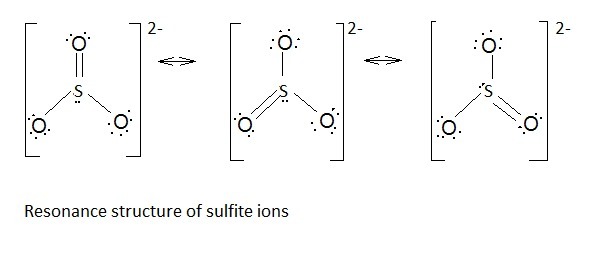
Sulphur has six valence electrons. One electron is shared by each oxide ion and two electrons are shared by oxygen atom and one electron pair is left unshared.
Thus, resonating structures are drawn when one structure cannot explain all the properties of the molecule.
Chapter 12 Solutions
World of Chemistry, 3rd edition
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





