
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 11.5, Problem 10ATS
Interpretation Introduction
Interpretation: Synthetic pathway of each of these compounds using
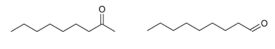
Concept introduction: One of the products is an
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
10.
M and N are amines with the molecular formula C3H»N. Reaction of M with sodium
nitrite and HCI releases nitrogen gas and produces a mixture of X and Y and Propanol
while N produces a yellowish oily compound, S when reacted with the same reagents.
Give the structures of M, N, X, Y, and S. Outline the synthesis of M from a suitable
alkene.
We discussed a tetrahedral intermediate in the context of reactions of derivatives of
carboxylic acids and nucleophilic substitution reactions. In all of these cases, the
tetrahedral intermediate was described as an unstable structure that undergoes
subsequent reactions. Briefly explain why tetrahedral intermediates are unstable.
1. Methylene cyclohexane, on treatment with strong acid, isomerises to yield methyl cyclohexene.
Propose a mechanism by which the reaction might occur.
Chapter 11 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Ch. 11.1 - Prob. 1CCCh. 11.1 - Prob. 2CCCh. 11.2 - Prob. 1LTSCh. 11.2 - Prob. 3PTSCh. 11.3 - Prob. 2LTSCh. 11.3 - Prob. 6ATSCh. 11.4 - Prob. 3LTSCh. 11.4 - Prob. 7PTSCh. 11.4 - Prob. 8ATSCh. 11.5 - Prob. 4LTS
Ch. 11.5 - Propose an efficient synthesis for each of the...Ch. 11.5 - Prob. 10ATSCh. 11 - Prob. 11PPCh. 11 - Prob. 12PPCh. 11 - Prob. 13PPCh. 11 - Prob. 14PPCh. 11 - Prob. 15PPCh. 11 - Prob. 16PPCh. 11 - Prob. 17PPCh. 11 - Prob. 18PPCh. 11 - Prob. 19PPCh. 11 - Prob. 20PPCh. 11 - Prob. 21PPCh. 11 - Prob. 22PPCh. 11 - Prob. 23PPCh. 11 - Prob. 24PPCh. 11 - Prob. 25PPCh. 11 - Prob. 26ASPCh. 11 - Prob. 27ASPCh. 11 - Prob. 28ASPCh. 11 - Prob. 29ASPCh. 11 - Prob. 30ASPCh. 11 - Prob. 31ASPCh. 11 - Prob. 32ASPCh. 11 - Prob. 33IPCh. 11 - Prob. 34IPCh. 11 - Prob. 35IPCh. 11 - Prob. 36IPCh. 11 - Prob. 37IPCh. 11 - Prob. 38IPCh. 11 - Prob. 39IPCh. 11 - Prob. 42CPCh. 11 - The compound Z-3-hexenyl acetate is one of several...Ch. 11 - When a consumer purchases a tomato, smell is one...Ch. 11 - Prob. 45CPCh. 11 - Prob. 46CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardThe following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forwardAlcohols are important for organic synthesis, especially in situations involving alkenes. The alcohol might be the desired product, or the OH group might be transformed into another functional group via halogenation, oxidation, or perhaps conversion to a sulfonic ester derivative. Formation of an alcohol from an alkene is particularly powerful because conditions can be chosen to produce either the Markovnikov or non-Markovnikov product from an unsymmetrical alkene. Using your reaction roadmap as a guide, show how to convert 4-methyl-1-pentene into 5-methylhexanenitrile. You must use 4-methyl-1-pentene and sodium cyanide as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.arrow_forward
- Reaction of -pinene with borane followed by treatment of the resulting trialkylborane with alkaline hydrogen peroxide gives the following alcohol. Of the four possible cis,trans isomers, one is formed in over 85% yield. (a) Draw structural formulas for the four possible cis,trans isomers of the bicyclic alcohol. (b) Which is the structure of the isomer formed in 85% yield? How do you account for its formation? Create a model to help you make this prediction.arrow_forwardThree isomeric pentanols with unbranched carbon chains exist. Which of these isomers, upon dehydration at 180C, yields only 1-pentene as a product?arrow_forwardA problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate: Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:arrow_forward
- Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardAnother important pattern in organic synthesis is the construction of CC bonds. Using your reaction roadmap as a guide, show how to convert propane into hex-1-en-4-yne. You must use propane as the source of all of the carbon atoms in the hex-1-en-4-yne product. Show all reagents needed and all molecules synthesized along the way.arrow_forwardGrignard reagent is a versatile tool in synthetic organic chemistry. Using bromocyclopentane as a starting material, show how a Grignard reagent, X, is synthesized. Reaction of X with water produces compound Y while treatment in carbon dioxide followed by hydrolysis forms compound Z. 3-methyl-2butanone reacts with X and hydrolyses to yield compound AA. Draw the structural formulae of compounds Y, Z and AA and write the chemical equations respectively.arrow_forward
- 10. MM and NN are amines with molecular formula, C3H9N. Reaction of MM with sodium nitrite and HCl releases nitrogen gas and produces a mixture of XX and YY, while NN produces yellowish oily compound ZZ when reacts with the same reagent. Gives the structures of MM, NN, XX, YY and ZZ. Outline a synthesis of MM from a suitable alkene.arrow_forwardThe structure below is the cyclic ether (epoxide), butene oxide: (1) CH3CH₂ -CH₂ butene oxide How could this compound be prepared from but-1-ene? Explain why butene oxide is much more reactive than its isomer, tetrahydrofuran, which is also a cyclic ether: H₂C-CH₂ H₂C CH₂ tetrahydrofuran Illustrate how butene oxide reacts with ammonia, showing details of the mechanism leading to the final product, C4H11 NO.arrow_forwardChemistry Provide reagents/conditions to accomplish the following syntheses. Several steps are required in some cases.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,