
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 11.11, Problem 45P
The following three nitrogen mustards were studied for possible clinical use. One is now used clinically, one was found to be too unreactive, and one was found to be too insoluble in water to be injected intravenously. Which is which? (Hint: Draw resonance contributors.)
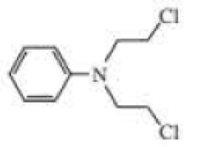
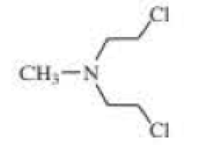
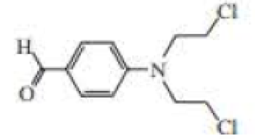
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Methyl acetate (CH;COOCH3) is formed by the reaction
of acetic acid with methyl alcohol. If the methyl alcohol
is labeled with oxygen-18, the oxygen-18 ends up in the
methyl acetate:
CH;COH + HSOCH3
CH;Č18OCH3 + H,O
(a) Do the C-OH bond of the acid and the O-H bond
of the alcohol break in the reaction, or do the O-H bond
of the acid and the C-OH bond of the alcohol break?
(b) Imagine a similar experiment using the radioisotope H,
which is called tritium and is usually denoted T. Would the
reaction between CH;COOH and TOCH; provide the same
information about which bond is broken as does the above
experiment with Hl$OCH;?
Consider the properties of sodium carbonate (Na2CO3), potassium carbonate (K2CO3), andsodium hydroxide (NaOH). How do they differ by pKa? Nucleophilicity? Solubility?
Despite its toxicity, arsenic is an essential
element. If humans do not consume at least
40 micrograms of this element per day it will
have adverse health effects. Natural medicine
sometimes offers arsenic supplements that
may contain the compounds shown below.
How would you synthesize the following
arsenic-based particles? HINT: start off with
yellow arsenic, As4, in each case!
a) ASF4(+)
b) ASF4(-)
c) AsO2(+)
d) ASO2F2(-)
Chapter 11 Solutions
Organic Chemistry
Ch. 11.1 - Why are NH3 and CH3NH2 no longer nucleophiles when...Ch. 11.1 - Prob. 2PCh. 11.1 - Prob. 5PCh. 11.2 - Prob. 7PCh. 11.3 - Prob. 9PCh. 11.3 - Show how 1-propanol can be converted into the...Ch. 11.4 - Which of the following alcohols dehydrates the...Ch. 11.4 - Prob. 12PCh. 11.4 - Prob. 13PCh. 11.4 - Propose a mechanism for each of the following...
Ch. 11.4 - Draw the product of each of the following...Ch. 11.4 - Explain why the following alcohols, when heated...Ch. 11.4 - What stereoisomers are formed in the following...Ch. 11.4 - Prob. 18PCh. 11.4 - What alcohol would you treat with phosphorus...Ch. 11.5 - Prob. 20PCh. 11.6 - Prob. 22PCh. 11.7 - Prob. 24PCh. 11.7 - Would you expect the reactivity of a five-membered...Ch. 11.7 - Prob. 26PCh. 11.7 - What products are obtained from the reaction of...Ch. 11.7 - Prob. 28PCh. 11.7 - Prob. 29PCh. 11.7 - Prob. 30PCh. 11.8 - Prob. 31PCh. 11.8 - Prob. 32PCh. 11.8 - How do the major products obtained from...Ch. 11.8 - Explain why the two arene oxides in Problem 38...Ch. 11.8 - Three arene oxides can be obtained from...Ch. 11.9 - Explain why the half-life (the time it takes for...Ch. 11.10 - Prob. 38PCh. 11.10 - Prob. 39PCh. 11.10 - Prob. 40PCh. 11.10 - Prob. 41PCh. 11.10 - Prob. 42PCh. 11.11 - Using an alkyl halide and a thiol as starting...Ch. 11.11 - The following three nitrogen mustards were studied...Ch. 11.11 - Why is melphalan a good cancer drug?Ch. 11.11 - Prob. 47PCh. 11 - Prob. 48PCh. 11 - Which compound is more likely to be carcinogenic?Ch. 11 - Prob. 50PCh. 11 - Prob. 51PCh. 11 - Write the appropriate reagent over each arrow.Ch. 11 - What alkenes would you expect to be obtained from...Ch. 11 - Prob. 54PCh. 11 - When heated with H2SO4, both...Ch. 11 - What is the major product obtained from the...Ch. 11 - When deuterated phenanthrene oxide undergoes a...Ch. 11 - An unknown alcohol with a molecular formula of...Ch. 11 - Prob. 59PCh. 11 - Prob. 60PCh. 11 - Propose a mechanism for the following reaction:Ch. 11 - What product would be formed if the four-membered...Ch. 11 - Which of the following ethers would be obtained in...Ch. 11 - Using the given starting material any necessary...Ch. 11 - Prob. 65PCh. 11 - When 3-methyl-2-butanol is heated with...Ch. 11 - Propose a mechanism for each of the following...Ch. 11 - How could you synthesize isopropyl propyl ether,...Ch. 11 - When the following seven-membered ring alcohol is...Ch. 11 - Ethylene oxide reacts readily with HO because of...Ch. 11 - Prob. 71PCh. 11 - Propose a mechanism for each of the following...Ch. 11 - Explain why the acid-catalyzed dehydration of an...Ch. 11 - Triethylene glycol is one of the products obtained...Ch. 11 - Prob. 75PCh. 11 - Prob. 76PCh. 11 - When ethyl ether is heated with excess HI for...Ch. 11 - Propose a mechanism for the following reaction:Ch. 11 - Prob. 79PCh. 11 - An ion with a positively charged nitrogen atom in...Ch. 11 - Propose a mechanism for each of the following...Ch. 11 - Prob. 82PCh. 11 - The following reaction takes place several times...Ch. 11 - A vicinal diol has OH groups on adjacent carbons....Ch. 11 - Prob. 85PCh. 11 - Prob. 86PCh. 11 - Two stereoisomers are obtained from the reaction...Ch. 11 - Propose a mechanism for each or the following...Ch. 11 - Triethylenemelamine (TEM) is an antitumor agent....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Selective serotonin reuptake inhibitors (SSRIs) are commonly prescribed antidepressants. SSRIs block the reabsorption of the neurotransmitter serotonin in the brain. Changing the balance of serotonin helps brain cells send and receive chemical messages, which in turn boosts mood. Two SSRI medications are Faverin (fluvoxamine, C15H21F3N2O2 ) and Zoloft (sertraline, C17H17Cl2N ). Determine the molar masses of Faverin and Zoloftarrow_forwardWhich of the following would effectively deprotonate an acid with pKa of 30?arrow_forward(i)What class of drug is Ranitidine? (ii)If water contains dissolved Ca2+ ions, out of soaps and synthetic detergents, which will you use for cleaning clothes? (iii)Which of the following is an antiseptic? 0.2% phenol, 1% phenolarrow_forward
- benzoic acid reacts with 3M NaOH, what does the final structure look like? (it is now a salt )arrow_forwardWhy is methyl salicylate so easily absorbed through the skin?arrow_forwardIn accordance to Dyuklo-Traube's rule in a homologous series of carboxylic acids, alcohols, amines at extension of length of the hydrocarbonic chain by one CH2-group the surface activity of substances is Select one: a. increased about 3-3,5 times b. increased about 0.5-1.5 times С. decreased about 30 times d. decreased about 2-3,5 times е. increased about 15 timesarrow_forward
- (d) What are the main hazards of disperse blue 14? (e) What are the main hazards of quinazarin green?arrow_forwardConjugate acid base pKa 2) Consider the reaction B: () + H3O+ →>> BH(+) + H2O. For the following named bases: 1) draw the structure of the base, 2) draw the structure of the conjugate acid, 3) give the name of the conjugate acid, and 4) give the approximate pKa of the conjugate acid (±1 pKa unit). For the purposes of answering this question you may use pKa values evenly divisible by 5. t-Butoxide anion p-Toluenesulfonate anion Triethylamine t-Butanol pKa pKa pKaarrow_forwardwhy do these compounds show this trend in pka?arrow_forward
- 4. Which effect does an antagonistic drug have? (a) It inhibits the activity of an enzyme (b) It promotes the activity of an enzyme (c) It inhibits the signalling of a receptor (d) It promotes the signalling of a receptor (e) It promotes nerve signalling 7. Which technique is not used for structural analyses of possible lead compounds? (a) NMR (b) IR (c) X-Ray crystallography (d) MRI (e) MSarrow_forwardMost of the pKa values given in this text were determined in water. How would the pKa values of carboxylic acids, alcohols, ammonium ions (RNH32) , phenol, and an anilinium ion (C6H5NH32) change if they were determined in a solvent less polar than water?arrow_forwardWhat is the dominant form of arsenic acid (H3A) at pH =9.23 ? Given: pKa1 = 2.24 , pKa2 = 6.96, pKa3 = 11.5arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
SAR of Anticancer(Antineoplastic) Drug/ Alkylating agents/ Nitrogen Mustard; Author: Pharmacy Lectures;https://www.youtube.com/watch?v=zrzyK3LhUXs;License: Standard YouTube License, CC-BY