
(a)
Interpretation : The change in pressure needs to be determined.
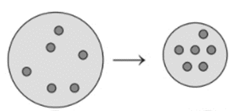
Concept Introduction : The ideal gas equation is combination of different
Here, P is pressure, V is volume, n is number of moles, R is Universal gas constant and T is temperature.
(b)
Interpretation : The change in pressure needs to be determined.
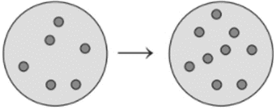
Concept Introduction : The ideal gas equation is combination of different gas laws such as Boyle’s law, Charles’s law, Gay-Lussac law etc. The combination of all these laws gives the ideal gas equation.
Here, P is pressure, V is volume, n is number of moles, R is Universal gas constant and T is temperature.
(c)
Interpretation : The change in pressure needs to be determined.
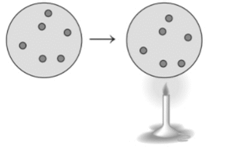
Concept Introduction : The ideal gas equation is combination of different gas laws such as Boyle’s law, Charles’s law, Gay-Lussac law etc. The combination of all these laws gives the ideal gas equation.
Here, P is pressure, V is volume, n is number of moles, R is Universal gas constant and T is temperature.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Basic Chemistry
- 28 A of ice at 0.0 °C is to a of at 8 A 45-g piece of ice at 0.0 °C is added to a sample of water at 8.0 °C. All of the ice melts and the temperature of the water decreases to 0.0 °C. How many grams of water were in the sample? (3.6, 3.7) oil in kcal/g? (3.5, 3.6) 9 In a large building, oil is used in a steam boiler heating system. The combustion of 1.0 lb of oil provides 2.4 × 107 J. (3.4, 3.6) a. How many kilograms of oil are needed to heat 150 kg of water from 22 °C to 100 °C? b. How many kilograms of oil are needed to change 150 kg of water to steam at 100 °C? 00 When 1.0 g of gasoline burns, it releases 11 kcal. The density of gasoline is 0.74 g/mL. (3.4, 3.6) a. How many megajoules are released when 1.0 gal of gaso- line burns? b. If a television requires 150 kJ/h to run, how many hours can the television run on the energy provided by 1.0 gal of gasoline? ISWERS wers to Selected Practice Problems a. element b. compound d. compound c. element e. compound a. pure substancearrow_forward(Q24) What is the density (in g/L) of sulfur dioxide gas at a pressure of 873 torr and a temperature of 69.0 °C? (3 sf)arrow_forwarddoes it compare with the known melting and boiling point? (1) 9. The average kinetic energy of water molecules is a measure of the temperature of water. When the temperature of water remains constant the average kinetic energy of the molecules remains constant, even though the water is being heated by the Bunsen flame. So, energy is being taken in by the water, but it is not being used to increase the kinetic energy of the molecules. 9.1 What type of energy are the water molecules gaining during a phase change? (1) 9.2 Explain your reasoning (to question 9.1) with reference to the kinetic theory of matter. 10. Write the conclusion. (3) [30]arrow_forward
- 10. 11.arrow_forward(6.5)What is the molar volume of argon gas at STP? O 15.3 L O 22.4 L O 4.00 L O 1.00 Larrow_forward(7.50ml) Charles' Law 1. Several balloons are inflated with helium to a volume of 0.75 L at 27°C. One of the balloons was found several hours later, the temperature had dropped to 22°C. What would be the volume of the balloon when found, if no helium has escaped? (0.74L) Sign out V 8 12 DELL %23 %24 & 4 6 [O e r t y u d. f m alt ctri ofarrow_forward
- 170 Given the following data: 0% 3% 16% 3% AH=+180.7 kJ AH=-113.1 kJ AH=-163.2 kJ O2(g) → 2NO(g), N2(g) + 2NO(g) + %3D 02(g) 2NO2(g), Sugera 52% 2N20(g) → 2N2(g) + O2(g), use Hess's law to calculate AH for the following reaction: N PLACE N20(g) + NO2(g)→3NO(g) Express the enthalpy in kilojoules to four significant digits. ? kJ Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining Provide Feedback O Esc DII PrtScn F1 F2 F3 F4 F5 F6 F7 F8 ! # 24 & 1 2 3 5 7 8. W Y 00 IIarrow_forwardL. Using Table 1, calculate AH; for each of the following reactions. Tell if each is exothermic or endothermic. a. 2 NO(g) + 0:(8) → 2 NO:(g) b. 2 C,Hlg) + 70:(g) → 4 CO.(g) + 6 H;0(g) c. N:(g) + 3 H:(g) - 2 NH:(g) d. 2 CH;OH(1) + 3 0:(g) → 2 CO:(g) + 4 H;0(g) e. 2 SO:(g) + 0:(8) + 2 S0,(g) f. 2 CH«(g) + 30:(8) → 2 CO:(g) + 2 H;0(g) & 2 CO(g) + O:(8) → 2 CO:(g) h. N;(8) + O:[g) → 2 NO(8) II. Calculate AH, for each of the following (using Table 1). i CHxo from the reaction 2 C;H:(B) + 5 0:(g) → 4 CO:(g) + 2 H;0(g) + 600kcal i CHo from the reaction CH«(g) • 2 0:(g) CO:(g) + 2 H;0(g) + 192 kcal III. Given the following reactions k S+ 0: - So: 71.0 kcal 2 so, + 0: - 2 s0, + 47.0 kcal Calculate AH, for 2 S + 3 0: - 2 SO, L 2C + 0, - 2 co + 52.8 kcal 2 co + 0; - 2 CO; + 125 kcal Calculate AlH, for C + 0z - co: IV. Calculate AH, AS and AG at 25 C, and tell if the reaction will go spontaneously (use Table 1 and 2) m. N:(g) + 3 H:(R) → 2 NH:(8) n. 2 CO(g) + 0:(g) - 2 CO:(8) a 2 CH:(g) + 5 0:(8)…arrow_forwardJania.20 Chapter 3 3.100 When 1.0 9 of gasoline burns, it releases gasoline 0.749/ML SI (3.4.3.6) A) HOw many megajoules arc released srs 1.0 gai ot gasoline burns? Whenarrow_forward
- H. C2H2 6. Draw out the Lewis structure for each. Determine the electron and molecular group geometry for each structure. Determine the intermolecular forces for each structure. (2pts) CF2H2 7. Circle the H-bonds on the following molecule. (1 pts) H H H H H 8. We experienced a large temperature swing during a softball weekend. If your tire pressure in the morning was 25 psi at -100C, what is the tire pressure at 40oC at 10 pm when you finish your last game? (1pt) 9. At the softball game, the caffeine in my Coke Zero was 46 mg for a 12 oz can. What is the ppm of caffeine in my Coke Zero? (2 pts)arrow_forward1. (9 points) A 15.0 gram piece of metal is heated to 65.0°C and then dropped into 175 grams of 24.0°C water. The system eventually comes to a stable temperature of 35.5 C. (The specific heat of water is 4.184 J/g °C) a. Energy is absorbed by the water. What is qWATER? b. Energy is released by the metal. What is qMETAL? (this one is easy) c. What is the specific heat (Cs) of the metal?arrow_forward(6.5:Similar to For More Practice 6.7) Density of a gas is 0.8916 g/L at 1.0 atm and 273 °C. Identify the gas. (Hint: Find the molar mass of the gas.) O oxygen (O₂) O chlorine gas (Cl₂) O argon (Ar) O none of thesearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





