
Basic Chemistry
6th Edition
ISBN: 9780134878119
Author: Timberlake, Karen C. , William
Publisher: Pearson,
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 11, Problem 85UTC
(a)
Interpretation Introduction
Interpretation:
From the given diagram, the gas sample at lowest pressure needs to be determined.
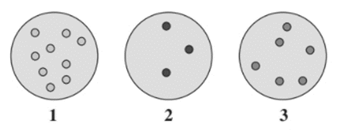
Concept Introduction :
The pressure depends on the number of gaseous molecules in a sample and space between them.
(b)
Interpretation Introduction
Interpretation:
From the given diagram, the gas sample at highest pressure needs to be determined.
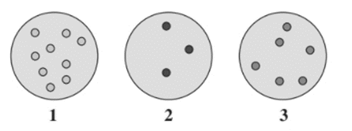
Concept Introduction :
The pressure depends on the number of gaseous molecules in a sample and space between them.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
320
29. Determine what happens to each variable below
(increases, decreases, no change) under the stated
conditions. Assume that all other variables not
specifically mentioned are constant values. (10.4)
(a) What happens to the pressure when the
volume decreases?
(b) What happens to the volume when the
temperature increases?
(c) What happens to the pressure when the
temperature decreases?
(d) What happens to the pressure when the
amount of gas decreases?
30. Determine what happens to each variable below
(increases, decreases, no change) under the stated
conditions. Assume that all other variables not
specifically mentioned are constant values. (10.4)
(a) What happens to the volume if the amount
of gas doubles?
(b) What happens to the temperature when the
pressure decreases?
(c) What happens to the temperature when the
volume increases?
34. Calculate the missing variables in each
experiment below using Boyle's law. (10.5)
(a) P, = 1.67 atm, V1 = 3.77 L, P2 = 0.229 atm,
V2 = ?
(b) P1 =…
The raté of effusion of a certain gas. was measured in a laboratory
Pure oxygen in the same
9.
apparatus and found to be 10:0 ml/sec.
apparatus at the same temperature and pressure effused at a rate of
Calculate the molecular weight of the gas. (63.6)
14.1 ml/sec.
ortant
(6.3)A gas occupies 50.0 L at 1525 mmHg. What volume (in L) will it occupy at 755 mmHg? Answer with 3 significant figures.
L
Chapter 11 Solutions
Basic Chemistry
Ch. 11.1 - Prob. 1PPCh. 11.1 - Prob. 2PPCh. 11.1 - Prob. 3PPCh. 11.1 - Prob. 4PPCh. 11.1 - Prob. 5PPCh. 11.1 - Prob. 6PPCh. 11.1 - Prob. 7PPCh. 11.1 - Prob. 8PPCh. 11.2 - Prob. 9PPCh. 11.2 - Prob. 10PP
Ch. 11.2 - Prob. 11PPCh. 11.2 - Prob. 12PPCh. 11.2 - Prob. 13PPCh. 11.2 - Prob. 14PPCh. 11.2 - Prob. 15PPCh. 11.2 - Prob. 16PPCh. 11.2 - Prob. 17PPCh. 11.2 - Prob. 18PPCh. 11.2 - Prob. 19PPCh. 11.2 - Prob. 20PPCh. 11.2 - Prob. 21PPCh. 11.2 - Prob. 22PPCh. 11.2 - Prob. 23PPCh. 11.2 - Prob. 24PPCh. 11.3 - Prob. 25PPCh. 11.3 - Prob. 26PPCh. 11.3 - Prob. 27PPCh. 11.3 - Prob. 28PPCh. 11.3 - Prob. 29PPCh. 11.3 - Prob. 30PPCh. 11.3 - Prob. 31PPCh. 11.3 - Prob. 32PPCh. 11.4 - Prob. 33PPCh. 11.4 - Prob. 34PPCh. 11.4 - Prob. 35PPCh. 11.4 - Prob. 36PPCh. 11.4 - Prob. 37PPCh. 11.4 - Prob. 38PPCh. 11.4 - Prob. 39PPCh. 11.4 - Explain each of the following observations: a....Ch. 11.4 - A tank contains isoflurane, an inhaled anesthetic,...Ch. 11.4 - Prob. 42PPCh. 11.5 - Prob. 43PPCh. 11.5 - Prob. 44PPCh. 11.5 - Prob. 45PPCh. 11.5 - Prob. 46PPCh. 11.5 - Prob. 47PPCh. 11.5 - Prob. 48PPCh. 11.6 - Prob. 49PPCh. 11.6 - Prob. 50PPCh. 11.6 - Prob. 51PPCh. 11.6 - Prob. 52PPCh. 11.6 - Prob. 53PPCh. 11.6 - Prob. 54PPCh. 11.7 - Prob. 55PPCh. 11.7 - What is the volume, in liters, of 4.00 mol of...Ch. 11.7 - An oxygen gas container has a volume of 20.0 L....Ch. 11.7 - Prob. 58PPCh. 11.7 - A 25.0-g sample of nitrogen, N2 , has a volume of...Ch. 11.7 - A 0.226-g sample of carbon dioxide, CO2 , has a...Ch. 11.7 - Prob. 61PPCh. 11.7 - Prob. 62PPCh. 11.7 - Prob. 63PPCh. 11.7 - Prob. 64PPCh. 11.8 - HCl reacts with magnesium metal to produce...Ch. 11.8 - When heated to 350Cat0.950atm , ammonium nitrate...Ch. 11.8 - Butane undergoes combustion when it reacts with...Ch. 11.8 - Potassium nitrate decomposes to potassium nitrite...Ch. 11.8 - Prob. 69PPCh. 11.8 - Nitrogen dioxide reacts with water to produce...Ch. 11.9 - Prob. 71PPCh. 11.9 - Prob. 72PPCh. 11.9 - Prob. 73PPCh. 11.9 - Prob. 74PPCh. 11.9 - Prob. 75PPCh. 11.9 - Prob. 76PPCh. 11.9 - An air sample in the lungs contains oxygen at 93...Ch. 11.9 - Prob. 78PPCh. 11.9 - Prob. 79PPCh. 11.9 - Prob. 80PPCh. 11.9 - Prob. 81PPCh. 11.9 - Prob. 82PPCh. 11 - Prob. 83UTCCh. 11 - Prob. 84UTCCh. 11 - Prob. 85UTCCh. 11 - Indicate which diagram (1, 2, or 3) represents the...Ch. 11 - A balloon is filled with helium gas with a partial...Ch. 11 - Prob. 88UTCCh. 11 - Prob. 89APPCh. 11 - In the fermentation of glucose (wine making), 780...Ch. 11 - Prob. 91APPCh. 11 - Prob. 92APPCh. 11 - In 1783, Jacques Charles launched his first...Ch. 11 - Prob. 94APPCh. 11 - Prob. 95APPCh. 11 - Prob. 96APPCh. 11 - Prob. 97APPCh. 11 - A steel cylinder with a volume of 15.0 L is filled...Ch. 11 - A sample of gas with a mass of 1.62 g occupies a...Ch. 11 - Prob. 100APPCh. 11 - How many grams of...Ch. 11 - A container is filled with...Ch. 11 - How many liters of H2 gas can be produced at...Ch. 11 - Prob. 104APPCh. 11 - Prob. 105APPCh. 11 - Hydrogen gas can be produced in the laboratory...Ch. 11 - Prob. 107APPCh. 11 - Prob. 108APPCh. 11 - A gas mixture contains oxygen and argon at partial...Ch. 11 - Prob. 110APPCh. 11 - Prob. 111CPCh. 11 - When heated, KClO3 forms KCl and O2 . When a...Ch. 11 - A sample of gas with a mass of 1.020 g occupies a...Ch. 11 - A sample of an unknown gas with a mass of 3.24 g...Ch. 11 - Prob. 115CPCh. 11 - When sensors in a car detect a collision, they...Ch. 11 - Prob. 117CPCh. 11 - Prob. 118CPCh. 11 - Prob. 119CPCh. 11 - A hyperbaric chamber has a volume of 1510 L. How...Ch. 11 - Laparoscopic surgery involves inflating the...Ch. 11 - Prob. 122CPCh. 11 - Prob. 123CPCh. 11 - Prob. 124CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 3. A fixed quantity of gas at 23.0°C exhibits a pressure of 748 torr and occupies a volume of 10.3 L. Calculate the volume the gas will occupy if the temperature is increased to 145°C while the pressure is held constant. (14.5L) Sign out DELL 5 alt ctriarrow_forward(7.50ml) Charles' Law 1. Several balloons are inflated with helium to a volume of 0.75 L at 27°C. One of the balloons was found several hours later, the temperature had dropped to 22°C. What would be the volume of the balloon when found, if no helium has escaped? (0.74L) Sign out V 8 12 DELL %23 %24 & 4 6 [O e r t y u d. f m alt ctri ofarrow_forward6. V₁6.00L P₁=730 P₂=760, (760.) (.) Calculate the final pressure inside a scuba tank after it cools from 1.00 x 10³ °C to 25.0 °C. The initial pressure in the tank is 130.0 atm. (6.00, 7. A sample of ammonia has a volume of 60.0 mL at 0°C. At what temperature will it have a volume of 325 mL? 60 77731arrow_forward
- 5. (8) A small bubble rises from the bottom of a lake, where the temperature and pressure are 7°C and 2.8 atm, to the water's surface, where the temperature is 27°C and pressure is 740 mmHg. Calculate the final volume in mL. of the bubble if it's initial volume was 1.5 mL.(706 mmHg = latm)arrow_forward14. Consider a container with a volume of 22.4 L filled with a gas at 1.00 atm and 0˚C. What will be the new pressure if the temperature increases to 298K? (1.09)arrow_forward(Q22) What will the pressure (in atm) when 61.9 milligrams of neon gas is held at a volume of 534 mL and a temperature of 41.8 °C? (3 sf)arrow_forward
- Fill in the blanks: (3.8) What volume (in mL) will 10.0 g of CO2 (g) Occupy at 27 °C and 2.00 atm? Atomic mass: C 12.01 O 16.00 Analysis (Given): T (CO2) = | K n mol (CO2) = %3D moL (3 sig fig) Answer: V (CO2) = mL %3D (3 sig fig)arrow_forward9.3 The balanced equation below represents the chemical reaction, where 3 moles of sodium react with chlorine. 2Na + Cl2 → 2NaCt 9.3.1 Calculate the mass of sodium chloride formed. 9.3.2 Calculate the volume of chlorine at STP if it reacts with 3,5 moles of sodium.arrow_forwardFill in the blanks: (6.5) The volume of a sample of gas at 50 °C is 2.50 L. Assume that the pressure is held constant. (a) What is the volume (in L) of the gas at -10. °C? (b). At what temperature (in °C) would the volume be 1.25 L? (c). At what temperature (in °C) would the volume be 2.75 L? Answer: (a) Volume at -10. °C = L (3 sig fig) (b) Temperature = °C (3 sig fig) (c) Temperature = °C (3 sig fig)arrow_forward
- (6.5)What is the molar volume of argon gas at STP? O 15.3 L O 22.4 L O 4.00 L O 1.00 Larrow_forward(6.6: Similar to For practice 6.10) The mixture of gas contains 3.5 g Ne and 2.5 g He. What is the total pressure be for the partial pressure of Ne to be 0.25 atm? O 1.5 atm O 0.43 atm O 2.1 atm O 1.2 atmarrow_forwardTwo (2.00) moles of a gas at 18°C occupies a volume of 5.00 L. a. What is the pressure of this gas sample? b. Calculate the new pressure of the gas when it is heated to 50° C.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY