
Concept explainers
Answer the following questions about erlotinib and phomallenic acid C. Erlotinib, sold under the trade name Tarceva, was introduced in 2004 for the treatment of lung cancer. Phomallenic acid C is an inhibitor of bacterial fatty acid synthesis.
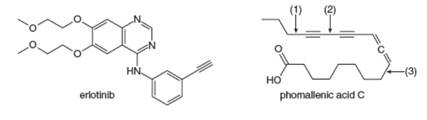
a. Which
b. What orbitals are used to form the shortest
c. Which H atom in phomallenic acid C is most acidic.
d. How many
e. Rank the labeled bonds in phomallenic acid C in order of increasing bond strength.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Organic Chemistry (6th Edition)
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
General, Organic, & Biological Chemistry
Chemistry
Chemistry In Context
Elementary Principles of Chemical Processes, Binder Ready Version
Fundamentals of Heat and Mass Transfer
- Atenolol is a β (beta) blocker, a drug used to treat high blood pressure. Which of the indicated N—H bonds is more acidic? Explain your reasoning.arrow_forwardWhich member of each pair is the stronger base? a. ethylamine or aniline b. ethylamine or ethoxide ion c. phenolate ion or ethoxide ion d. phenolate ion or acetate ionarrow_forward4-Methylphenol is more acidic than ethanol (pKa 10.36 vs 16.0) , even though both contain an OH group and a methyl group. Draw the structures of the anions formed from loss of the alcoholic protons from both compounds. Use resonance to explain the difference in their respective acidities.arrow_forward
- Explain why m-nitroaniline is a stronger base than p-nitroaniline.arrow_forwardOH Xou XCOOH X CN COOH B C D 3) In which set are the compounds arranged in order of decreasing acidity? A. 2-bromopropanoic acid>propanoic acid>propanol B. propanoic acid> propanol>2-bromopropanoic acid C. 2,2,2-trifluoroethanoic acid >2-bromopropanoic acid>propanol D. propanol>propanoic acid>2-bromopropanoic acid -CN Abo OH NH₂arrow_forward7. Draw the conjugate base for each. CH;CH2OH → CH:CH;CH3 → CH:CH2NH2 – CH:CH2SH –arrow_forward
- НО. meta-Hydroxyacetophenone HO para-Hydroxyacetophenone The conjugate base of the para isomer has a negative charge that is delocalized over atom(s), and the conjugate base of the meta isomer has a negative charge that is delocalized over oxygen atom(s). Therefore, the conjugate base of the meta isomer is carbon atom(s) and isomer, making meta-hydroxyacetophenone oxygen carbon atom(s) and stable than then conjugate base of the para ✓acidic than para-hydroxyacetophenone.arrow_forwardRank the nitrogen atoms in each compound in order of increasing basicity. Isoniazid is a drug used to treat tuberculosis, whereas histamine (Section 25.6B) causes the runny nose and watery eyes associated with allergies. NH2 NHNH2 b. а. isoniazid histaminearrow_forwardPhenol has a pka = 10.0. 4-Hydroxybenzaldehyde has a pka = 7.3. Draw resonance forms of the conjugate base of both and explain why 4-hydroxybenzaldehyde is more acidic. (Acidic H is underlined.) OH phenol OH -H* - H* 4-hyroxybenzaldehydearrow_forward
- 1. Consider the following unequal equilibrium. Which two species in this equilibrium represent the weaker acid and the weaker base? CH3CH3 + CH3NH 1 2 O 1 and 4 2 and 3 1 and 2 3 and 4 ? → CH3CH₂ 3 CH3NH₂ 4arrow_forwardWhich is the more stable base? a. Br− or I− b. CH3O− or CH3S− c. CH3CH2O− or CH3COO− d. H2C CH or HC C− e. FCH2CH2COO− or CH2CH2COO- f. CLCH2CH2O- or CL2CHCH2O-arrow_forwardNH2 .NH2 Rank the compounds in order of increasing basicity. .NH2 NH2 O2N بھی کبک سبک کیک C D A B Darrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



