
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 7E
Label each atom marked with an arrow with the appropriate shape name, and estimate the bond angles around it as being close to one of 180°, 120°, 109.5° or 90°. (circled charges indicate the charge on the molecule or fragment)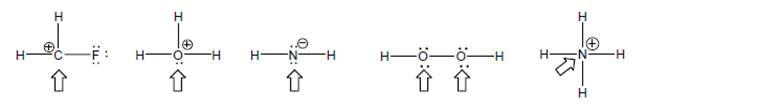
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw out the organic shorthand for the molecules below, and then draw another possible resonance structure of the same molecule next to with arrows to show electron movement.
-CH3CH2CONH3
-CH3CH2COOH
Make a model of propane (C3H8), and draw this model using dashed lines and wedges to represent bonds going back and coming forward
Rank the molecules in order of polarity from least to most polar. Justify your answer.
NH3 , CH3CL ,H20, CCL4
Chapter 1 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 1 - (E) What does the number (+Z) at the center of...Ch. 1 - Prob. 2CTQCh. 1 - Prob. 3CTQCh. 1 - Prob. 4CTQCh. 1 - Prob. 5CTQCh. 1 - Prob. 6CTQCh. 1 - Prob. 7CTQCh. 1 - You hear a student from a nearby group say that...Ch. 1 - Use VSEPR to explain why the HBH bond angle of BH3...Ch. 1 - Both the HCH and HCO bond angles of H2CO...
Ch. 1 - Prob. 11CTQCh. 1 - Consider the following flat drawing of methane...Ch. 1 - Use VSEPR to assign a value of (close to) 109.5,...Ch. 1 - A student draws the picture of ammonia (NH3) in...Ch. 1 - Prob. 15CTQCh. 1 - How many central atoms does the molecule H2NCH3...Ch. 1 - Indicate the bond angle and shape about each...Ch. 1 - Explain how there can be two kinds of bent:...Ch. 1 - A student makes the following statement: “The...Ch. 1 - A student who missed this class needs to know how...Ch. 1 - Prob. 1ECh. 1 - Prob. 2ECh. 1 - Consider the incomplete valence shell...Ch. 1 - How many valence electrons does a neutral a. K...Ch. 1 - Consider the molecules AlCl3 (aluminum chloride)...Ch. 1 - Draw an example of a bent molecule with a bond...Ch. 1 - Label each atom marked with an arrow with the...Ch. 1 - a model of each of the following molecules: a....
Additional Science Textbook Solutions
Find more solutions based on key concepts
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
The structural formula of 1, 2-dimethylbenzene needs to be drawn. Concept introduction: The ring structures of ...
Chemistry: Matter and Change
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Rank the following bonds from most polar to least polar?Cl-O, Mg-O, O-O, C-Oarrow_forwardAdd lone pairs where needed to give octets and then determine the shape around each indicated atom.arrow_forwardThe curved arrow notation introduced in Section 1.6B is a powerful method used by organic chemists to show the movement of electrons not only in resonance structures, but also in chemical reactions.Because each curved arrow shows the movement of two electrons, following the curved arrows illustrates what bonds are broken and formed in a reaction. Consider the following three-step process. (a) Add curved arrows in Step [1] to show the movement of electrons. (b) Use the curved arrows drawn in Step [2] to identify the structure of X. X is converted in Step [3] to phenol and HCl.arrow_forward
- Which of the following has the most polar indicated bond (Electronegativites are not necesasry to answer this question)? a. Si-H b. P-H c. S-H d. Cl-Harrow_forwardHow do you know when to draw a solid wedge vs a dashed wedge when drawing 3D bond-line structures? I know that solid-wedge means the atom is pointing towards you and dashed wedge means it's in the back, but how do you know which atoms are in the front as opposed to the back? How can you tell what the configuration will look like in space just by looking at the lewis structure or name?arrow_forwardDraw the BEST lewis structure (with formal charges) of the indicated chemical species. (Draw all possible resonance structures). 1. HBrO2 2. BrO3^-arrow_forward
- Please answer this question, Dont use cursive handwriting, answer in clear step by step solutions, For the following molecules, give the: Lewis diagram, name of shape, bond angles (DON'T use bond angles from vsepr chart), and polarity a) F2O b) SF6 Thank you!arrow_forwardAnswer the questions based on the resonance structures of S2O32– (thiosulfate) below. See attatched photo for more information. Draw an “X” through the resonance structure that is not chemically possible. On the remaining three resonance structures, write in any non-zero formal charges. Of the remaining three structures, circle the structure that will contribute the least to the resonance hybrid. Draw a resonance hybrid (including partial “δ” and full charges) for S2O32–.arrow_forwardFor each the following molecules, indicate the direction of each bond polarity using arrows AND also indicate if the overall structure is polar or non- polar.arrow_forward
- 1) Draw the lewis structure in bond line notation, with correct bond geometry (include formal charge if necessary) 2) What is the geometry of the carbon highlighted in bold? 3) What is the bond angle of carbon?arrow_forwardCreatine is a dietary supplement used by some athletes to boost theirathletic performance. (a) Draw in all lone pairs in creatine. (b) Draw two additional resonance structures showing all lone pairs and formal charges.arrow_forwardAnswer the following true or false questions about the molecule acrylaldehyde, its resonance structures, and resonance hybrid. In fairness, many of these statements are false and represent common misconceptions about resonance. If a statement is false as written, change the underlined fragment of the statement to make it true. \ 1) Atoms can be moved around to make a new resonance form 2) The oxygen atom in acrylaldehyde has a full negative charge 3) These three resonance structures exist at different points in timearrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER


World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY