
Chemistry: The Molecular Science
5th Edition
ISBN: 9781285199047
Author: John W. Moore, Conrad L. Stanitski
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
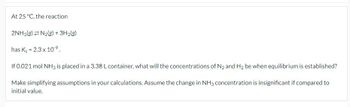
Transcribed Image Text:At 25 °C, the reaction
2NH3(g) N2(g) + 3H2(g)
has Kc = 2.3 x 10-9.
If 0.021 mol NH3 is placed in a 3.38 L container, what will the concentrations of N2 and H2 be when equilibrium is established?
Make simplifying assumptions in your calculations. Assume the change in NH3 concentration is insignificant if compared to
initial value.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Similar questions
- Write an equation for an equilibrium system that would lead to the following expressions (ac) for K. (a) K=(Pco)2 (PH2)5(PC2H6)(PH2O)2 (b) K=(PNH3)4 (PO2)5(PNO)4 (PH2O)6 (c) K=[ ClO3 ]2 [ Mn2+ ]2(Pcl2)[ MNO4 ]2 [ H+ ]4 ; liquid water is a productarrow_forwardWhat is the law of mass action? Is it true that the value of K depends on the amounts of reactants and products mixed together initially? Explain. Is it true that reactions with large equilibrium constant values are very fast? Explain. There is only one value of the equilibrium constant for a particular system at a particular temperature, but there is an infinite number of equilibrium positions. Explain.arrow_forwardSuppose a reaction has the equilibrium constant K = 1.3 108. What does the magnitude of this constant tell you about the relative concentrations of products and reactants that will be present once equilibrium is reached? Is this reaction likely to be a good source of the products?arrow_forward
- Write a chemical equation for an equilibrium system that would lead to the following expressions (ad) for K. (a) K=(PH2S)2 (PO2)3(PSO2)2 (PH2O)2 (b) K=(PF2)1/2 (PI2)1/2PIF (c) K=[ Cl ]2(Pcl2)[ Br ]2 (d) K=(PNO)2 (PH2O)4 [ Cu2+ ]3[ NO3 ]2 [ H+ ]8arrow_forwardBecause calcium carbonate is a sink for CO32- in a lake, the student in Exercise 12.39 decides to go a step further and examine the equilibrium between carbonate ion and CaCOj. The reaction is Ca2+(aq) + COj2_(aq) ** CaCO,(s) The equilibrium constant for this reaction is 2.1 X 10*. If the initial calcium ion concentration is 0.02 AI and the carbonate concentration is 0.03 AI, what are the equilibrium concentrations of the ions? A student is simulating the carbonic acid—hydrogen carbonate equilibrium in a lake: H2COj(aq) H+(aq) + HCO}‘(aq) K = 4.4 X 10"7 She starts with 0.1000 AI carbonic acid. What are the concentrations of all species at equilibrium?arrow_forwardFor the reactionH2(g)+I2(g)2HI(g), consider two possibilities: (a) you mix 0.5 mole of each reactant. allow the system to come to equilibrium, and then add another mole of H2 and allow the system to reach equilibrium again. or (b) you mix 1.5 moles of H2 and 0.5 mole of I2 and allow the system to reach equilibrium. Will the final equilibrium mixture be different for the two procedures? Explain.arrow_forward
- . What does it mean to say that a state of chemical or physical equilibrium is dynamic?arrow_forwardConsider 0.200 mol phosphorus pentachloride sealed in a 2.0-L container at 620 K. The equilibrium constant, Kc, is 0.60 for PCl5(g) PCl3(g) + Cl2(g) Calculate the concentrations of all species after equilibrium has been reached.arrow_forwardWrite equilibrium constant expressions for the following generalized reactions. a. 2X(g)+3Y(g)2Z(g) b. 2X(g)+3Y(s)2Z(g) c. 2X(s)+3Y(s)2Z(g) d. 2X(g)+3Y(g)2Z(s)arrow_forward
- Explain that equilibrium is dynamic, and that at equilibrium the forward and backward reaction rates are equal.arrow_forwardIn a solution with carbon tetrachloride as the solvent, the compound VCl4. undergoes dimerization: 2VCl4V2Cl8 When 6.6834 g VCl4. is dissolved in 100.0 g carbon tetrachloride, the freezing point is lowered by 5.97C. Calculate the value of the equilibrium constant for the dimerization of VCl4 at this temperature. (The density of the equilibrium mixture is 1.696 g/cm3, and Kf = 29.8C kg/mol for CCl4.)arrow_forwardShow that the complete chemical equation, the total ionic equation, and the net ionic equation for the reaction represented by the equation KI(aq)+I2(aq)KI3(aq) give the same expression for the reaction quotient. KI3 is composed of the ions K+ and I3-.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning
