
Concept explainers
Name the following
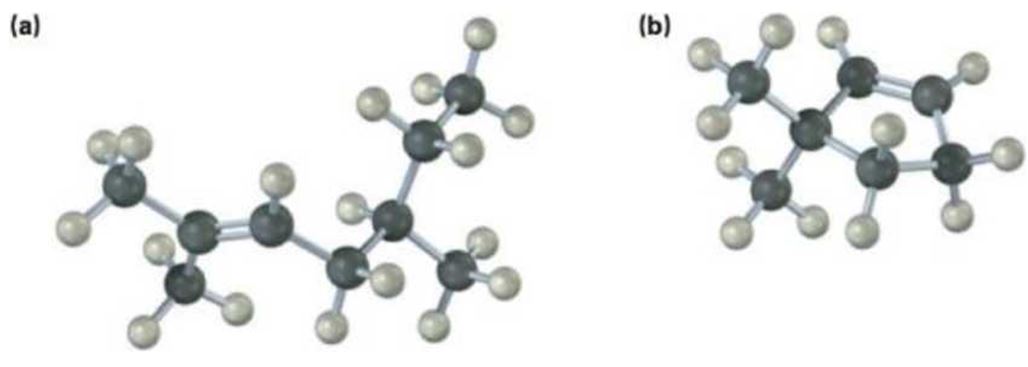
a)
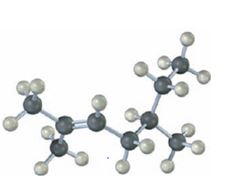
Interpretation:
The name of the alkene shown to be given. The products of its reaction with 1) meta-chloroperoxybenzoic acid, 2) KMnO4 in aqueous acid and 3) O3, followed by Zn in acetic acid, are to be given.
Concept introduction:
When alkenes react with meta-chloroperoxybenzoic acid, oxygen atom adds to the double bond to give epoxides.
Upon treatment with KMnO4 in aqueous acid, if the alkene the double bonded carbon is di-substituted and has no hydrogen it is converted in to a ketone and if the double bonded carbon is mono-substituted and has one hydrogen it is oxidized to a carboxylic acid.
Upon treatment with O3, followed by Zn in acetic acid the double bond is cleaved and each carbon in the double bond gets attached to an oxygen atom to give carbonyl compounds as products. If in the alkene double bonded carbon is di-substituted and has no hydrogen it is converted in to a ketone and if the double bonded carbon is mono-substituted and has one hydrogen it is oxidized to an aldehyde.
To give:
The name of the alkene shown and the products of its reaction with 1) meta-chloroperoxybenzoic acid, 2) KMnO4 in aqueous acid and 3) O3, followed by Zn in acetic acid.
Answer to Problem 22VC
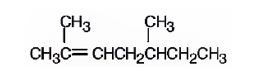
The name of the alkene shown is 2,5-dimethyl-2-heptene.
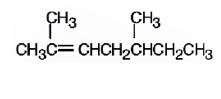
The product formed when it reacts with meta-chloroperoxybenzoic acid is
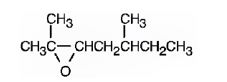
The products formed when it reacts with KMnO4 in aqueous acid are
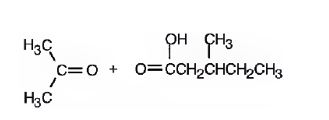
The products formed when it reacts with O3, followed by Zn in acetic acid are
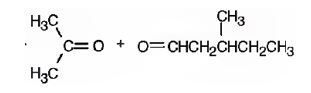
Explanation of Solution
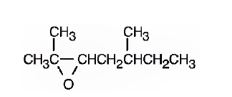
2,5-dimethyl-2-heptene has a double bond between C2&C3. When treated with meta-chloroperoxybenzoic acid, oxygen atom adds to both C2&C3 to give an epoxide.
In 2,5-dimethyl-2-heptene, C2 has no hydrogens attached. So it is oxidized to a ketone.C3 has one hydrogen and is oxidized to a carboxylic acid.
Upon treatment with O3, followed by Zn in acetic acid the double bond between C2&C3 in 2,5-dimethyl-2-heptene is cleaved and each carbon gets attached to an oxygen atom to give a ketone and aldehyde as products.
The name of the alkene shown is 2,5-dimethyl-2-heptene is
The product formed when it reacts with meta-chloroperoxybenzoic acid is
The products formed when it reacts with KMnO4 in aqueous acid are
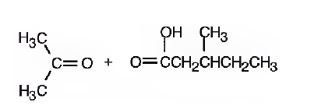
The products formed when it reacts with O3, followed by Zn in acetic acid are
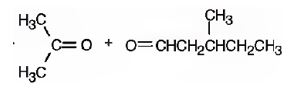
b)
Interpretation:
The name of the alkene shown to be given. The products of its reaction with 1) meta-chloroperoxybenzoic acid, 2) KMnO4 in aqueous acid and 3) O3, followed by Zn in acetic acid, are to be given.
Concept introduction:
When alkenes react with meta-chloroperoxybenzoic acid, oxygen atom adds to the double bond to give epoxides.
Upon treatment with KMnO4 in aqueous acid, if in the alkene double bonded carbon is di-substituted and has no hydrogen it is converted in to a ketone and if the double bonded carbon is mono-substituted and has one hydrogen it is oxidized to a carboxylic acid.
Upon treatment with O3, followed by Zn in acetic acid the double bonded is cleaved and each carbon in the double bond gets attached to an oxygen atom to give carbonyl compounds as products. If in the alkene double bonded carbon is di-substituted and has no hydrogen it is converted in to a ketone and if the double bonded carbon is mono-substituted and has one hydrogen it is oxidized to an aldehyde.
To give:
The name of the alkene shown and the products of its reaction with 1) meta-chloroperoxybenzoic acid, 2) KMnO4 in aqueous acid and 3) O3, followed by Zn in acetic acid.
Answer to Problem 22VC
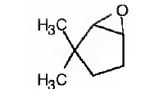
The name of the alkene shown is 3,3-dimethylcyclopentene.
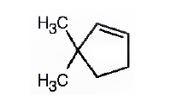
The product formed when it reacts with meta-chloroperoxybenzoic acid is
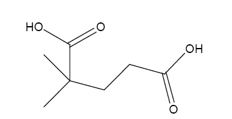
The products formed when it reacts with KMnO4 in aqueous acid are
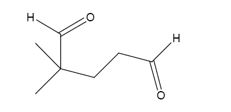
The products formed when it reacts with O3, followed by Zn in acetic acid are
Explanation of Solution
3,3-dimethylcyclopentene has a double bond between C1&C2. When treated with meta-chloroperoxybenzoic acid, oxygen atom adds to both C1&C2 to give an epoxide.
In 3,3-dimethylcyclopentene both C1&C2 have one hydrogen attached to them. So they are oxidized to carboxylic acids when treated with KMnO4.
Upon treatment with O3, followed by Zn in acetic acid the double bond between C1&C2 in 3,3-dimethylcyclopentene is cleaved and each carbon with one hydrogen gets attached to an oxygen atom to yield a dialdehyde as product.
The name of the alkene shown is 3,3-dimethylcyclopentene.

The products formed when it reacts with meta-chloroperoxybenzoic acid are

The products formed when it reacts with KMnO4 in aqueous acid are

The products formed when it reacts with O3, followed by Zn in acetic acid are

Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry
- What is the name of the following molecule Ph-CH2-CH2-CH2-CH=CH2? * styrene 5-phenyl-1-PENTENE 1-phenyl-4-PENTENE 3-benzyl-1-BUTENE allylbenzenearrow_forwardReaction of 2-methylpropene with CH3OH in the presence of H2SO4 catalyst yieldsmethyl tert-butyl ether, CH3OC(CH3)3, by a mechanism analogous to that of acid- catalyzed alkene hydration. Write the mechanism, using curved arrows for each step.arrow_forwardWhat is the name of the product obtained by the reaction of iso-butylbenzene with KMN04/HO-? Name the product of the following reaction: KMNO4 H,0 O,Narrow_forward
- Na, NH3 H3C-C=C-CH3 CH3 H3C° Alkynes are reduced to trans alkenes by a process called dissolving metal reduction. The reaction uses sodium or lithium metal as the reducing agent and liquid ammonia as the solvent. The method is specific in the formation of trans alkenes from alkynes. The method involves two successive transfers of single electrons from the alkali metal to the triple bond, with abstraction of protons from the ammonia solvent. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions .CH3 H-NH2 CH3 :NH2 H3C H3Carrow_forwardWhat alkene with the molecular formula C6H12, when treated with ozone and then dimethyl sulfide, gives the following product(s)?arrow_forwardWhat alkene is needed to synthesize each 1,2-diol using [1] OsO4 followed by NaHSO3 in H2O; or [2] CH3CO3H followed by −OH in H2O?arrow_forward
- Alloocimene is a hydrocarbon found in turpentine. It has the molecular formula C₁0H16 and a UV absorption maximum at 290 nm. On hydrogenation with a palladium catalyst, 2,6-dimethyloctane is obtained. Ozonolysis of alloocimene, followed by treatment with zinc and acetic acid, produces the following four fragments: || CH3C-CH i O || || CH3CCH3 HC-CH Propose a structure for alloocimene. O || CH3CHarrow_forwardA compound with formula C7H12O is treated with sodium borohydride in methanol to yield 2,2-dimethylcylopentanol. Write a reaction scheme showing the structures of the reactant, the reagents, and the product. Will the product be optically active? Explain.arrow_forwardPlease give the main substitution product for each of the following reactions, and indicate the dominant mechanism: (a) 1-bromopropane + NaOCH3 → (b) 3-bromo-3-methylpentane + NaOC2H5 →arrow_forward
- 5. Compound A, C 10H 18O, undergoes reaction with dilute H 2SO 4 at 50 °C to yield a mixture of two alkenes, C 10H 16. The major alkene B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Which of the following reactions are correct.arrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardPropose a synthesis for (Z)-9-tricosene (muscalure), the sex pheromone for the common housefly (Musca domestica), starting with acetylene and haloalkanes as sources of carbon atoms.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

