
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 7, Problem 16CTQ
Interpretation Introduction
Interpretation: In the given structures, each black ball needs to be labeled as up or down.
Concept Introduction: Chair conformation is the most stable conformation of cyclohexane. It is represented as follows:

Here, the substituted groups in the chair conformation are represented as follows:
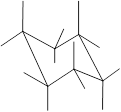
The groups showing in straight upward and downward directions are axial and the groups bend slightly right or left are equatorial.
The axial and equatorial groups are represented in the chair conformation as A and E:
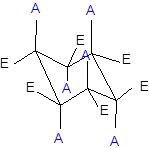
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. Start with a 7 carbon chain.
Add 2 methyl groups at the 2nd and 4th carbon.
Add an alcohol group at the 2nd carbon.
Add an additional 4 carbon chain coming off the
6th carbon.
4. Start with NH3 and show step-by-step how the compound N-sec-butyl-N-isopropyl-3-pentanamine can be
formed. Draw and name the reactants and products in each reaction (3 separate reactions.)
5. Salicylic acid (shown below) is a polyfunctional molecule (contains more than one functional group.) Circle and
identify the functional groups on salicylic acid. Salicylic acid can react with ethanoic acid (acetic acid) to form
aspirin. It can also react with ethanol to form ethyl salicylate, a compound with a minty smell used in perfume
and artificial flavors. Draw out structures to illustrate each of these reactions. Discuss how these two reactions
are similar and how they are different.
HO,
HO
I need help on letter 4E only. I drew the structural form already*
Chapter 7 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 7 - Prob. 1CTQCh. 7 - Prob. 2CTQCh. 7 - Prob. 3CTQCh. 7 - Draw wedge and dash skeletal representations of...Ch. 7 - Label each ring in Figure 7.2 cis or trans.Ch. 7 - Prob. 6CTQCh. 7 - Prob. 7CTQCh. 7 - Prob. 8CTQCh. 7 - a model of cyclohexane in a chair conformation,...Ch. 7 - Prob. 10CTQ
Ch. 7 - Prob. 11CTQCh. 7 - Fill in the blanks: cis-1,3-Dimethylcyclohexane...Ch. 7 - Prob. 13CTQCh. 7 - Prob. 14CTQCh. 7 - Prob. 15CTQCh. 7 - Prob. 16CTQCh. 7 - Prob. 17CTQCh. 7 - Prob. 18CTQCh. 7 - Draw chair representations of...Ch. 7 - Which stereoisomer in the previous question is...Ch. 7 - Prob. 21CTQCh. 7 - Prob. 1ECh. 7 - Label each of the following as cis, trans or...Ch. 7 - Which pair has more in common with one another?Ch. 7 - Prob. 4ECh. 7 - Prob. 5ECh. 7 - Prob. 6ECh. 7 - Fill in the table by drawing a representation of a...Ch. 7 - Prob. 9ECh. 7 - True or False: If you perform a chair flip on...Ch. 7 - Prob. 11ECh. 7 - Prob. 12ECh. 7 - Prob. 13ECh. 7 - Prob. 14ECh. 7 - Prob. 15ECh. 7 - Draw trans-1-tert-butyl-3-methylcyclohexane in its...Ch. 7 - Build a model of methylcyclohexane, and use the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Replace one H on propane with Br. Does it matter which H is replaced? Yes or No (circle) Are isomers possible for C3H¬B1? Yes or No (circle) Draw the expanded structures for all isomers and name each compound. As in question 1 on the previous page, use a number in front of the name to indicate the position for the bromine atom on the carbon chain. Consult with your instructor to make sure you've correctly drawn and named the structure(s) here. 3. Complete the following for compounds derived from butane, C4H10. Butane, C4H10 Lewis Structures & Name Are isomers possible? Yes or No (circle) Build a model & draw the line structures for all potential structural isomers of C4H10. Using your molecular models for the butane isomers as a guide, draw line structures for all monoiodo-derivatives of each of your isomers of butane having the molecular formula C4H9I. Give the IUPAC name for each compound.arrow_forwardQuestion #2 - Start with a cyclohexane. Add a methyl to one of the carbons. Add a single oxygen to the carbon opposite the methyl. Add a 2 carbon chain to that lone oxygen.arrow_forwardLook at the third carbon in the longest carbon chain. To draw the molecule in 3D on a 2D surface (such as paper), the Br could have solid wedge showing that it projects to the back of the molecule (behind the paper/screen), and the methyl group could have a dashed wedge showing that it projects to the front of the molecule (out of the paper/screen). Br True Falsearrow_forward
- Zanamivir is an antiviral drug. Identify and circle each functional group (write for example primary amine, secondary alcohol, aldehyde, alkyne, etc.).arrow_forwardA student suggests that the molecule on the right can be made from a single molecule with one less ring in it. If the student is correct, draw the starting material below, otherwise, check the box under the drawing area. ? + NaOH Click and drag to start drawing a structure. olo Ararrow_forward20. Circle any two structures. Below each structure, write the name of the compound. Onl graded if you draw more than two. Use cis or trans in the name.arrow_forward
- View the first compound name provided in Table 6. Follow the steps below and draw each part of the structure on a piece of paper. Determine the number of carbons present in the compound based on the base name. Draw the carbon chain and include any double or triple bonds if indicated in the suffix of the base name. Number each carbon. The carbons can be numbered from left to right or right to left. Draw any substituents on the corresponding carbon atom for which is indicated in the name. Refer to Figures 3 and 4 in the background for a visual representation of numbered carbons with corresponding substituents. Check that each carbon atom has a total of 4 bonds.arrow_forward2. Consider the following organic molecules A-H. Many of them are natural products isolated from Nature. Please answer the following questions by writing the Letter pertaining to each molecule next to the question. -N- H;C "CO,H N. Но CH2 ČH3 B strychnine (a plant alkaloid) hirsutic acid A nepetalactone (essence of cutnip) (a fimgul metabolito) H- H. I CH,0 E helminthosporal (a imgal toxu) D progesterone F cantharidin (a progestin hommone) (am insect vesicant) Br NO2 Br CI G untenine C (E)-1,6-dibromo-2,7-dichloro-3,7-dimethyloct-3-ene (fom red algae) (4 sponge metabolite)arrow_forwardWhich is number 1 carbon and why? I thought it would be 1,2,4 but apparently it's 1,3,4 starting from the top right methyl group carbon.arrow_forward
- A student suggests that the molecule on the right can be made from a single molecule with one less ring in it. If the student is correct, draw the starting material below, otherwise, check the box under the drawing area. Click and drag to start drawing a structure. + NaOH C ? Ararrow_forward1. Does chlorophyll a or chlorophyll b have a greater number of “conjugated” double bonds, or do they have the same number? Draw the structure of chlorophyll b and using a colored highlighter, indicate which of the double bonds are “conjugated”. 2. What is the main functional group in THF (tetrahydrofuran)? Is THF more polar or less polar than IPA (also called: isopropyl alcohol, isopropanol, or 2-propanol)? 3.If you mistakenly used hexanes-THF (2:1) for eluting the carotene pigments from your chromatography column, will they elute faster or slower than if you had correctly used hexanes-THF (10:1)? Explainarrow_forwardBoxes 1-4box 1 answer choices: addition, condensation, substitution, or hydrolysis. box 2 answer choices: amide, alcohol, carboxylic acid, or side group. box 3 answer choices: alcohol, amine, amide, or ester. box 4 answer choices: ester, amide, carboxylic acid, or amine.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY