
(a)
Interpretation:
The reagent that is needed to convert to ![]() each of the compound needs to be identified.
each of the compound needs to be identified.
Concept introduction:
Addition of H2O to an
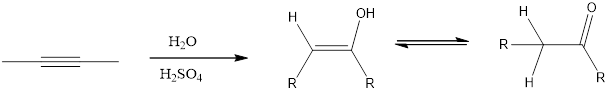
(b)
Interpretation:
The reagent that is needed to ![]() convert to each of the compound needs to be identified.
convert to each of the compound needs to be identified.
Concept introduction:
Addition of 1. R2BHand 2. H2O2, OH- reagent to an alkyne is a hydroboration-oxidation reaction to form the keto product as shown below in the fig. Addition of borane forms an organoborane. Oxidation with basic H20 2 forms an enol. Tautomerization of the enol forms a carbonyl compound. The overall result is addition of H2O to a triple bond.
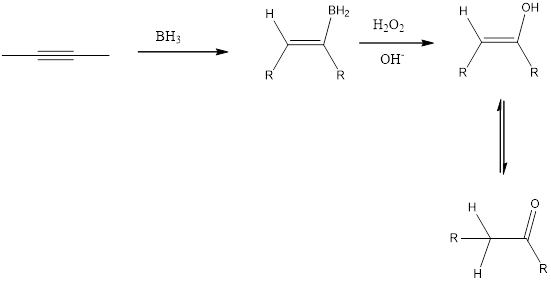
(c)
Interpretation:
The reagent that is needed to convert to ![]() each of the compound needs to be identified.
each of the compound needs to be identified.
Concept introduction:
Reaction between the 3-hexyne and HBr reagent is an example of electrophilic addition. The alkynes undergoes addition reaction because of pi bond in them. The electrophilic end of the reagent is attracted to the electron rich triple bond. It is a sequential reaction in which the addition of the reagent to alkyne to alkene which then addition of one more equivalent reagent to form the four new bond as
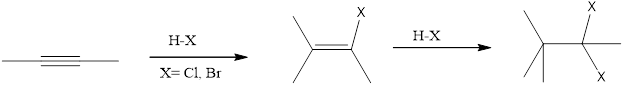
(d)
Interpretation:
The reagent that is needed to ![]() convert to each of the compound needs to be identified.
convert to each of the compound needs to be identified.
Concept introduction:
Terminal alkynes are readily converted into acetylide anions using the strong bases such as NaNH2 and NaH. These anions are strong nucleophiles, capable of reacting with electrophiles such as
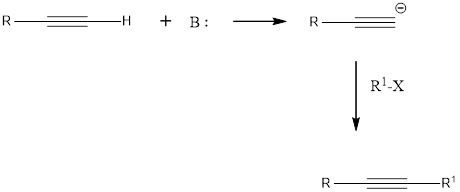
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Organic Chemistry
- 66. What reagents yield the desired product? Br a b. C d. 1) CH,CH2CH2 CH2CI, AICI 2) Br2, FeBry 1) CH3CH2CH2COCI, AICI3 2) NH2NH2, KOH, A 3) Br2, FoBr3 1) Br2, FeBr3 2) CH3CH2CH2COCI, AICI3 3) NH2NH2, KOH, A 1) CH3CH2CH2COCI, AICI, 2) Br2, FeBr3 3) NH2NH2, KOH, Aarrow_forwardWhat reagents are needed to convert each compound to benzaldehyde (CgHsCHO)? More than one step may be required. a. CgHsCH2OH b. CęHsCOCI c. CgHsCOOCH3 d. CęH;COOH e. CgHsCH3 f. CeHsCH=CH2 g. CeHsCH=NCH2CH;CH3 h. CgH;CH(OCH2CH32arrow_forwardThese reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O, HgSO4 choices:A,DB,CA,B,CA,B,C,Darrow_forward
- 3. Choose the appropriate reagent OH HO a. NaCN, then CO2 b. LIAIH4, then CO2 c. NACN, then H2O in HC1 d. LIAIH4, then H2O in HC1arrow_forwardGive the IUPAC name for each compound. a. (CHa)3CCH,CH(CHCH3)2 b. CH3(CH2)3CH(CH,CH2CH3)CH(CH32 d.arrow_forward5. What reagents are needed to convert toluene (C,H,CH,) to each compound? a. C.H.COOH b. C.H₂CH₂Br c. p-bromotoluene d. o-nitrotoluene e. p-ethyltoluene f.arrow_forward
- Draw the products formed when hex-1-yne is treated with each reagent. a. HCl (2 equiv) b. HBr (2 equiv) c. Cl2 (2 equiv) d.H2O + H2SO4 + HgSO4 e. [1] R2BH; [2] H2O2, HO− f. NaH g. [1] −NH2; [2] CH3CH2Brarrow_forwardDraw the products formed when phenol (C6H5OH) is treated with each set of reagents.a. [1] HNO3, H2SO4; [2] Sn, HCIb. [1] (CH3CH2)2CHCOCI, AlCl3; [2] Zn(Hg), HCIc. [1] CH3CH2CI, AlCl3; [2] Br2, hvd. [1] (CH3)2CHCI, AlCl3; [2] KMnO4arrow_forwardWhat crossed Claisen product is formed from each pair of compounds? a. CH,CH,COOE and HCO,Et b. CH3(CH2),CO,Et and HCO,Et c. (CH3),C= 0 and CH,CO,Et d. OEt andarrow_forward
- What alkenes are formed when each alcohol is dehydrated with TSOH? Label the major product when a mixture results. CH2CH3 OH он OH b. d. CH;CH2CH2CH2OH а. C. е. Онarrow_forwardWhich group in each pair is assigned the higher priority? a. – CH3, – CH2CH3 b. – I, – Br c. – H, – D d. – CH2Br, – CH2CH2Br e. – CH2CH2Cl, – CH2CH(CH3)2 f. – CH2OH, – CHOarrow_forwardWhat is the product of the following reaction? A. B. -N O A B O C CI OD CH3NH₂ (2 equiv) -NH₂ C. D. 11 ? NHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





