
Concept explainers
(a)
Interpretation:
The monomer required to produce the given polymer needs to be determined.
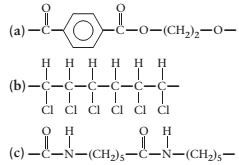
Concept Introduction:
A polymer is a long chain consists of large number of monomer units. The monomers are repeated units in a polymer they are linked to each other in different orders to produce different
These monomer units are linked to each other either through the formation of peptide linkage or glycosidic linkage or by removal of any moiety such as a water molecule.
Polyvinyl chloride, bakelite, polystyrene are some of the example of polymers.
(b)
Interpretation:
The monomer required to produce the given polymer needs to be determined.
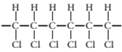
Concept Introduction:
A polymer is a long chain consists of large number of monomer units. The monomers are repeated units in a polymer they are linked to each other in different orders to produce different polymers.
These monomer units are linked to each other either through the formation of peptide linkage or glycosidic linkage or by removal of any moiety such as a water molecule.
Polyvinyl chloride, bakelite, polystyrene are some of the example of polymers.
(c)
Interpretation:
The monomer required to produce the given polymer needs to be determined.
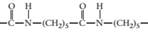
Concept Introduction:
A polymer is a long chain consists of large number of monomer units. The monomers are repeated units in a polymer they are linked to each other in different orders to produce different polymers.
These monomer units are linked to each other either through the formation of peptide linkage or glycosidic linkage or by removal of any moiety such as a water molecule.
Polyvinyl chloride, bakelite, polystyrene are some of the example of polymers.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Chemistry: Principles and Reactions
- What is the process by which polymers are broken down into monomers? What is added to the polymers during the reaction (what goes into the reaction?)arrow_forwardDraw the monomer that is used to create the following polymers.arrow_forwardWhich is one of the simplest synthetic polymers? How does it react?arrow_forward
- What is the structure of the monomer that forms this polymer?arrow_forwardQuestion 9: Draw the structure of the polymer formed from the addition of the following two monomers: HOOC -COOH + H₂N- O -NH₂arrow_forwardWhich monomer is used in the forming of the following polymer? A) I B) II C) III D) IVarrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
