
Concept explainers
(a)
Interpretation:
The monomer required to produce the given polymer needs to be determined.
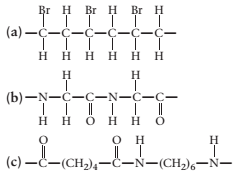
Concept Introduction:
A polymer is a long chain consists of large number of monomer units. The monomers are repeated units in a polymer they are linked to each other in different orders to produce different
These monomer units are linked to each other either through the formation of peptide linkage or glycosidic linkage or by removal of any moiety such as a water molecule.
Polyvinyl chloride, bakelite, polystyrene are some of the example of polymers.
(b)
Interpretation:
The monomer required to produce the given polymer needs to be determined.
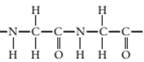
Concept Introduction:
A polymer is a long chain consists of large number of monomer units. The monomers are repeated units in a polymer they are linked to each other in different orders to produce different polymers.
These monomer units are linked to each other either through the formation of peptide linkage or glycosidic linkage or by removal of any moiety such as a water molecule.
Polyvinyl chloride, bakelite, polystyrene are some of the example of polymers.
(c)
Interpretation:
The monomer required to produce the given polymer needs to be determined.
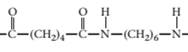
Concept Introduction:
A polymer is a long chain consists of large number of monomer units. The monomers are repeated units in a polymer they are linked to each other in different orders to produce different polymers.
These monomer units are linked to each other either through the formation of peptide linkage or glycosidic linkage or by removal of any moiety such as a water molecule.
Polyvinyl chloride, bakelite, polystyrene are some of the example of polymers.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Chemistry: Principles and Reactions
- What is the process by which monomers are joined together to form polymers? What is removed from the monomers during the reaction (what comes out of the reaction)?arrow_forwardComplete the polymersarrow_forwardWhat is the process by which polymers are broken down into monomers? What is added to the polymers during the reaction (what goes into the reaction?)arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





