
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 9.2, Problem 1LTS
Interpretation Introduction
Interpretation:
The systematic name of the given compound needs to be written down.
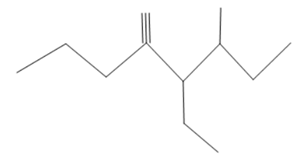
Concept Introduction :
Nomenclature is the process of giving distinctive names to chemical compounds so that they may be quickly distinguished as distinct chemicals.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Give a systematic name for compound 2.1 including its stereochemical designation
Compound B of molecular formula C9H19N shows a noteworthy infrared absorption at 3300 cm-1. Its 1H-NMR spectrum shows three singlets – δ 1.0 (6H), 1.1 (12H), 1.4 (1H) ppm. Its 13C-NMR spectrum has four signals – δ 25, 28, 41, 64 ppm. Suggest a structure for this compound.
Compound B has molecular formula C9H10. The IR spectrum is shown below. The 1H-NMR spectrum shows a multiplet at 7.2 ppm integrating to 4H, a triplet at 2.9 ppm integrating to 4H, and a triplet at 2.1 ppm integrating to 2 H. Suggest a structure for B and explain your reasoning
Chapter 9 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give IUPAC names for the following substances (red = O, blue = N):arrow_forwardCompound A of molecular formula C3H6O shows a noteworthy infrared absorption at 1716 cm-1. Its 1H-NMR spectrum shows one singlet – δ 2.2 (6H) ppm. Its 13C-NMR spectrum has two signals – δ 30, 207 ppm. Suggest a structure for this compound.arrow_forwardThe 1H-NMR spectrum of Compound D of molecular formula C10H12O shows three singlets – δ 2.20 (6H, s), 4.86 (4H), 7.10 (2H) ppm. Its 13C-NMR spectrum has five signals – 20, 74, 127, 135, 146 ppm. Suggest a structure for this compound.arrow_forward
- Compound A (C6H12O2) reacts with water, acid, and heatto yield compound B (C5H10O2) and compound C (CH4O).Compound B is acidic. Deduce possible structures of compounds A, B, and Carrow_forwardCompounds B and C are isomers with molecular formula C5H9BrO2. The 1H NMR spectrum of compounds B and C are shown below. The IR spectrum corresponding to compound B showed strong absorption bands at 1739, 1225, and 1158 cm-1, while the spectrum corresponding to compound C have strong bands at 1735, 1237, and 1182 cm-1. 1.Based on the information provided, determine the structure of compounds B and C. 2.Assign all peaks in 1H NMR spectrum of compounds B and C.arrow_forwardThe proton NMR spectrum of a compound with formula C8H14O2 is shown. TheDEPT experimental results are tabulated. The infrared spectrum showsmedium-sized bands at 3055, 2960, 2875, and 1660 cm-1and strong bands at1725 and 1185 cm-1. Draw the structure of this compound and assign thecarbon peaks.arrow_forward
- Provide structures for the following compoundsarrow_forwardPropose structures for molecules that fit the following descriptions:(a) An aldehyde with the formula C5H10O(b) An ester with the formula C6H12O2(c) A compound with the formula C3H7NOS that is both anamide and a thiolarrow_forwardUse the 1H NMR and IR spectra given below to identify the structure ofcompound B (molecular formula C4H8O2).arrow_forward
- A compound has the molecular formula C6H12O2. Its IR spectrum shows a strong absorption band near 1740 cm-1. Its 1H NMR spectrum consists of two singlets at δ 1.2 and δ 3.6. Which is the most likely structure of the compound?arrow_forwardGive IUPAC names for the following compounds:arrow_forwardDraw the structure of a compound, C4H8O3 that exhibits IR absorptions at 1710 and 2500-3000 cm-1 and thefollowing 1H NMR signals: 11.1 (1H, singlet), 4.14 (2H, singlet), 3.63 (2H, quartet), 1.26 (3H, triplet) ppm.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY