
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 81P
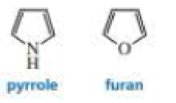
Why is the delocalization energy of pyrrole (21 kcal/mol) greater than that of furan (16 kcal/mol)?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Label the stereogenic center(s) in each drug as R or S. L-Dopa is used to treat Parkinson's disease (Chapter 1). Ketamine is an
anesthetic. Enalapril belongs to a class of drugs called ACE inhibitors, which are used to lower blood pressure.
CH3
NH CI
COOH
CH,CH,O,C
a.
Но
H NH2
b.
OH
enalapril
Trade name: Vasotec
L-dopa
ketamine
If a mixture has an ee of 90%, what is the percentage of each enantiomer?
Multiple Choice
80% : 20%
55% : 45%
95% : 5%
65% : 35%
Draw the product formed when pentanal (CH3 CH₂ CH₂ CH₂ CHO) is treated with each reagent. With some
reagents, no reaction occurs.
a. NaBH4, CH3OH
b. [1] LiAiH4: [2] H₂O
c. H₂, Pd-C
d. PCC
e. Na₂Cr₂O7, H₂SO4, H₂O
f. Ag₂O, NH4OH
g. [1] CH3 MgBr; [2] H₂O
h. [1] C6H5 Li: [2] H₂O
i. [1] (CH3)2 CuLi; [2] H₂O
J. [1] HC=CNa; [2] H₂O
k. [1] CH 3 C=CLI; [2] H₂O
1. The product in (a), then TBDMS-CI, imidazole
Chapter 8 Solutions
Organic Chemistry
Ch. 8.1 - Prob. 1PCh. 8.1 - Prob. 2PCh. 8.4 - Prob. 3PCh. 8.5 - Prob. 5PCh. 8.6 - a. Predict the relative bond lengths of the three...Ch. 8.6 - Prob. 7PCh. 8.6 - Prob. 8PCh. 8.8 - Prob. 9PCh. 8.9 - Prob. 10PCh. 8.9 - Prob. 12P
Ch. 8.9 - Prob. 13PCh. 8.10 - Prob. 14PCh. 8.10 - What orbitals contain the electrons represented as...Ch. 8.10 - Prob. 16PCh. 8.10 - Prob. 17PCh. 8.11 - Prob. 18PCh. 8.11 - Prob. 19PCh. 8.11 - Prob. 20PCh. 8.12 - Prob. 21PCh. 8.12 - Prob. 22PCh. 8.12 - Prob. 23PCh. 8.13 - Prob. 24PCh. 8.13 - Prob. 25PCh. 8.13 - Prob. 26PCh. 8.14 - Prob. 27PCh. 8.14 - Prob. 28PCh. 8.14 - Prob. 29PCh. 8.15 - Which member of each pair is the stronger acid?Ch. 8.15 - Which member of each pair is the stronger base? a....Ch. 8.15 - Rank the following compounds from strongest acid...Ch. 8.15 - Prob. 34PCh. 8.16 - Prob. 35PCh. 8.17 - Prob. 37PCh. 8.17 - Prob. 38PCh. 8.17 - Prob. 39PCh. 8.17 - Prob. 40PCh. 8.17 - Prob. 41PCh. 8.17 - Prob. 42PCh. 8.18 - Prob. 43PCh. 8.18 - Prob. 44PCh. 8.18 - Prob. 45PCh. 8.18 - Prob. 47PCh. 8.19 - Prob. 48PCh. 8.19 - Prob. 49PCh. 8.19 - Prob. 50PCh. 8.19 - Prob. 51PCh. 8.19 - Prob. 52PCh. 8.19 - Prob. 53PCh. 8.19 - Prob. 55PCh. 8.20 - Prob. 56PCh. 8 - Prob. 57PCh. 8 - Prob. 58PCh. 8 - Prob. 59PCh. 8 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - Prob. 65PCh. 8 - Prob. 66PCh. 8 - Prob. 67PCh. 8 - Prob. 68PCh. 8 - Prob. 69PCh. 8 - Which compound is the strongest base?Ch. 8 - Prob. 71PCh. 8 - Prob. 72PCh. 8 - Prob. 73PCh. 8 - Prob. 74PCh. 8 - Prob. 75PCh. 8 - Prob. 76PCh. 8 - Prob. 77PCh. 8 - Prob. 78PCh. 8 - Purine is a heterocyclic compound with four...Ch. 8 - Prob. 80PCh. 8 - Why is the delocalization energy of pyrrole (21...Ch. 8 - Prob. 82PCh. 8 - Prob. 83PCh. 8 - Prob. 84PCh. 8 - A student obtained two products from the reaction...Ch. 8 - Prob. 86PCh. 8 - a. How could each of the following compounds be...Ch. 8 - Draw the products obtained from the reaction of...Ch. 8 - How would the following substituents affect the...Ch. 8 - Prob. 90PCh. 8 - The acid dissociation constant (Ka) for loss of a...Ch. 8 - Protonated cyclohexylamine has a Ka = 1 1011...Ch. 8 - Prob. 93PCh. 8 - Prob. 94PCh. 8 - Prob. 95PCh. 8 - Prob. 96PCh. 8 - Prob. 97PCh. 8 - a. Propose n mechanism for the following reaction:...Ch. 8 - Prob. 99PCh. 8 - As many as 18 different Diels-Alder products can...Ch. 8 - Prob. 101PCh. 8 - Prob. 102PCh. 8 - Prob. 103PCh. 8 - Prob. 104PCh. 8 - The experiment shown next and discussed in Section...Ch. 8 - Prob. 106PCh. 8 - Prob. 107PCh. 8 - Prob. 108PCh. 8 - Prob. 1PCh. 8 - Prob. 2PCh. 8 - Prob. 3PCh. 8 - Prob. 4PCh. 8 - Prob. 5PCh. 8 - Prob. 6PCh. 8 - Prob. 7PCh. 8 - Prob. 8PCh. 8 - Prob. 9PCh. 8 - Prob. 10PCh. 8 - Prob. 11PCh. 8 - Prob. 12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Following are four stereo representations for lactic acid. Which are diastereomers? Hill H3C A COOH a. A and B OH HOW HOOC B CH 3 H b. B and C ww COOH нощу H C CH3 c. B and D M H HO D CH 3 COOH d. A and C ww e. None of thesearrow_forwardLabel the stereogenic center(s) in each drug as R or S. L-Dopa is used to treat Parkinson's disease (Chapter 1). Ketamine is an anesthetic. Enalapril belongs to a class of drugs called ACE inhibitors, which are used to lower blood pressure. CH3 NHẠC CH,CH,Oz a. HO OH COOH HÌNH, L-dopa b. ketamine H enalapril Trade name: Vasotec CO₂Harrow_forwardWhat is the structure of 2-trifluoromethyl-2- methoxybutanal? MeO I F₂C 11 CF₁ IV CF3arrow_forward
- Compare the reaction between 2,4,6-cycloheptatrienone and cyclopentadiene to the reaction between 2,4,6-cycloheptatrienone and ethene. Why does 2,4,6-cycloheptatrienone use two T electrons in one reaction and four T electrons in the other? a. b. + CH2=CH2arrow_forwardDraw the products formed when (S)-butan-2-ol is treated with TsCl and pyridine, followed by NaOH. Label the stereogenic center in each compound as R or S. What is the stereochemical relationship between the starting alcohol and the nal product?arrow_forwardLocate the stereogenic center in each compound and draw both enantiomers. b. HO, OH С. HO. NH2 a.arrow_forward
- Which statement is true about these Fischer structures? CH3 CH3 CH3 HO CH3 it: -H HO- -H H- -OH H- HO- -H- H- -OH H- HO- -- CH3 CH3 CH3 CH3 I II III IV O A I and II are identical (same stereoisomers). II and IV are identical. I and II are diastereoisomers. O D.II is a meso structure.arrow_forwardH H₂C Which statement is true about the Following pair of compounds? H₂C H COOH NH₂ H H H COOH NH₂ They have: a) same physical and chemical properties b) same chemical behavior towards chiral reagents c) different physical and chemical properties d) same chemical behavior towards achiral reagentsarrow_forwardWhat is the relationship between the parent structure on the left and the structure on the right? HO O O O COOH H H-OH B A: Enantiomers B: Diastereomers C: Completely unrelated molecules D: Identical molecules CH₂OH HO HO COOH H H CH₂OHarrow_forward
- Give the product for each step in the reactionarrow_forwardCelery ketone, like carvone, has two distinct aromas. In contrast to the S enantiomer's licorice scent, the R enantiomer has an earthy, celery-like aroma. Each enantiomer should be depicted and its odor assigned.arrow_forwardThe delocalization energy of an ester is about 10 kcal/mol, and the delocalization energy of an amide isabout 18 kcal/mol.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY