
Biochemistry
9th Edition
ISBN: 9781319114671
Author: Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Question
Chapter 8, Problem 49P
Interpretation Introduction
Interpretation:
The functions of the non-catalytic amino acids are to be stated.
Concept introduction:
The organic compounds that contain 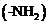 and carboxyl
and carboxyl 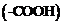
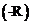 are known as amino acids. Proteins are the
are known as amino acids. Proteins are the
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
This image is showing the conformations of ubiquitin superimposed. What do the structures in this figure tell us about the dynamics of enzymes? Discuss the limitations of crystal structures in the analysis of proteins. What are the benefits? What are the advantages of NMR structures?
Wolfenden has stated that it is meaningless to distinguish between the “binding sites” and the “catalytic sites” of enzymes. Explain.
List three effects of macromolecular crowding on the properties of enzymes and the reactions they catalyze.
Chapter 8 Solutions
Biochemistry
Ch. 8 - Prob. 1PCh. 8 - Prob. 2PCh. 8 - Prob. 3PCh. 8 - Prob. 4PCh. 8 - Prob. 5PCh. 8 - Prob. 6PCh. 8 - Prob. 7PCh. 8 - Prob. 8PCh. 8 - Prob. 9PCh. 8 - Prob. 10P
Ch. 8 - Prob. 11PCh. 8 - Prob. 12PCh. 8 - Prob. 13PCh. 8 - Prob. 14PCh. 8 - Prob. 15PCh. 8 - Prob. 16PCh. 8 - Prob. 17PCh. 8 - Prob. 18PCh. 8 - Prob. 19PCh. 8 - Prob. 20PCh. 8 - Prob. 21PCh. 8 - Prob. 22PCh. 8 - Prob. 23PCh. 8 - Prob. 24PCh. 8 - Prob. 25PCh. 8 - Prob. 26PCh. 8 - Prob. 27PCh. 8 - Prob. 28PCh. 8 - Prob. 29PCh. 8 - Prob. 30PCh. 8 - Prob. 31PCh. 8 - Prob. 32PCh. 8 - Prob. 33PCh. 8 - Prob. 34PCh. 8 - Prob. 35PCh. 8 - Prob. 36PCh. 8 - Prob. 37PCh. 8 - Prob. 38PCh. 8 - Prob. 39PCh. 8 - Prob. 40PCh. 8 - Prob. 41PCh. 8 - Prob. 42PCh. 8 - Prob. 43PCh. 8 - Prob. 44PCh. 8 - Prob. 45PCh. 8 - Prob. 46PCh. 8 - Prob. 47PCh. 8 - Prob. 48PCh. 8 - Prob. 49P
Knowledge Booster
Similar questions
- True or False Immobilization improves the stability of the enzyme. EnaLne, has a half-life of 10 days in free solution, but under identical conditions of temperature, pH, and medium composition, the measured half-life of a packed column is 30 days. The enzyme is immobilized in a porous sphere 5 mm in diameter.arrow_forwardA generalized enzyme active site is shaped like a hemisphere with a radius of 45Å. The active site holds the following amino acids in a homeostatic solution (pH = 7.38): -HAVARILKHAVARILKHAVARILK- Assuming the charge is distributed uniformly along the hemisphere, determine the force at which this active site acts upon a single ATP molecule at the center of the hemisphere.arrow_forwardSeveral factors contribute to enzyme catalysis. What arethey? Briefly explain the effect of each.arrow_forward
- Explain why the very tight binding of a substrate to an enzyme is not desirable for enzyme catalysis, whereas tight binding of the transition state is desirable.arrow_forwardRuBP carboxylase is by no means an ideal enzyme. Describe some of the problems with its active site and its substrate specificity. If we compare the amino acid sequences of this enzyme from many different species, they are almost identical. What is the significance of this uniformity?arrow_forwardThe pKa for histidine is pKa = 6.1 while that for cysteine is pKa = 8.0 2. Assume that both histidine and cysteine are catalytic groups for a particular enzyme. Assume also that the side chain of cysteine must be in the deprotonated form. Estimate the pH at which the catalytic activity of this enzyme is the maximum and sketch a pH-activity graph.arrow_forward
- "Enzyme immobilization confers greater stability to the enzyme"-Do you agree or disagree with the statement? With appropriate example justify your opinion. Why the material used to immobilize the enzyme must be inert?arrow_forwardIn covalent catalysis, either nucleophilic catalysis or electrophilic catalysis occurs. Do you agree or disagree with this statement? Explain.arrow_forwardWhat happens to a denatured enzyme regarding its functionality? How can that result be explained with the help of the lock and key model?arrow_forward
- Enzymes are proteins that increase the rate of chemical reactions by lowering the energy activation required to facilitate the process. Explain the generalized and sequential enzyme mechanism of action (hint: from substrates to products).arrow_forwardThe breakdown of a complex carbohydrate into its monomeric unit can be described as an enzyme-catalyzed reaction that has a deltaG(∆G) of -90kcal/mol. Suppose you were to add triple the amount of enzyme into this reaction - what would be the ∆G of this reaction?arrow_forwardWhen enzyme solutions are heated, there is a progressive loss of catalytic activity over time due to denaturation of the enzyme. A solution of the enzyme hexokinase incubated at 450C lost 50% of its activity in 12 minutes, but when incubated at 450C in the presence of a very large concentration of one of its substrates, it lost only 3% of its activity in 12 minutes. Suggest why thermal denaturation of hexokinase was retarded in the presence of one substratesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON