
Interpretation:
The enol tautomers for the given
Concept Introduction:
Acid Catalysed addition of water: When water is added to alkyne in the presence of an acid, the product formed will be an enol. Enol contains a double bond and an
If a carbonyl group is bonded to two alkyl groups, it is called as a ketone. The enol formed in the acid catalysed addition of water will be easily converted into a ketone.
Conversion of terminal
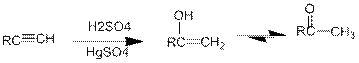
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry, Books a la Carte Edition (8th Edition)
- Draw the enol tautomers for each of the following compounds. For compounds that have more than one enol tautomer, indicate the one that is more stable.arrow_forwardDraw the structure of the major organic product for the following reaction. OH HO H₂SOarrow_forwardAddition of water to an alkyne gives a keto‑enol tautomer product. Draw the ketone that is in equilibrium with the given enol.arrow_forward
- The major product of the given reaction has the molecular formula CH03. Draw its structure in the most stable tautomeric form. Select Draw Rings More Erase H 1. NaOCH,CH3 2. H3O* CH;CH2OHarrow_forwardPropene undergoes tautomerization after hydroboration to create a more stable ketone product. True or Falsearrow_forwardTautomers are constitutional isomers that differ in the location of adouble bond and a hydrogen atom. Two tautomers are in equilibriumwith each other. Explain this ?arrow_forward
- Provide a complete step-by-step mechanism for the following reaction. ОН Conc. H2SO4 heatarrow_forwardDraw the product formed from cyclohex-2-enone in each of three reactions. Reaction 1 Reaction 2 Reaction 1 1. NaCN 1. (CH3)2CuLi Reaction 2 product product 2. H3O*, H20 2. CH3CH21 A 1. EtO,CCH,CO¿Et NaOEt, HOET 2. Hао", Н-0, д Reaction 3 Reaction 3 productarrow_forwardDraw the major organic substitution product for the reaction shown. он CH;CH2OH isopropyl alcohol Incorrectarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





