
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.1, Problem 1P
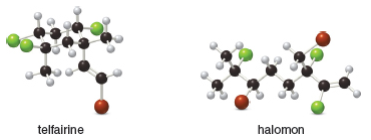
Telfairine, a naturally occurring insecticide, and halomon, an antitumor agent, are two
polyhalogenated compounds isolated from red algae. (a) Classify each halide bonded to an
hybridized carbon as 1°, 2°, or 3°. (b) Label each halide as vinyl, allylic, or neither.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the alkenes shown in the ball-and-stick models (A–C) in order of increasing stability.
Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation.
c. CH3CH2Cl, AlCl3
l. product in (c), then KMnO4
a. What five-carbon alkene forms the same product whether it reacts with HBr in the presence of a peroxide or with HBr in the absence of a peroxide?b. Draw the structures of four six-carbon alkenes that form the same product, whether they react with HBr in the presence of a peroxide or with HBr inthe absence of a peroxide.
Chapter 7 Solutions
Organic Chemistry (6th Edition)
Ch. 7.1 - Problem 7.1 Telfairine, a naturally occurring...Ch. 7.2 - Give the IUPAC name for each compound. a. b. c. d.Ch. 7.2 - Prob. 3PCh. 7.3 - An sp3 hybridized CCl bond is more polar than an...Ch. 7.4 - Prob. 5PCh. 7.6 - Prob. 6PCh. 7.7 - Prob. 10PCh. 7.8 - Prob. 15PCh. 7.8 - Prob. 16PCh. 7.8 - Prob. 17P
Ch. 7.11 - Prob. 18PCh. 7.11 - Prob. 19PCh. 7.11 - Draw the product of each SN2 reaction and indicate...Ch. 7.11 - Prob. 21PCh. 7.11 - Prob. 22PCh. 7.12 - What happens to the rate of an SN1 reaction under...Ch. 7.12 - Draw the products of each SN1 reaction and...Ch. 7.13 - Classify each carbocation as 1,2, or 3. a. b. c....Ch. 7.15 - Problem 7.30 For each alkyl halide and...Ch. 7.15 - Prob. 30PCh. 7.15 - Prob. 31PCh. 7.15 - Prob. 32PCh. 7.15 - Prob. 33PCh. 7.15 - Prob. 34PCh. 7 - Give the IUPAC name for each compound, including...Ch. 7 - Draw the products of each nucleophilic...Ch. 7 - Prob. 50PCh. 7 - Prob. 51PCh. 7 - 7.53 Consider the following reaction.
Draw a...Ch. 7 - Prob. 57PCh. 7 - Prob. 58PCh. 7 - Consider the following SN1 reaction. a.Draw a...Ch. 7 - 7.59 Pick the reactant or solvent in each part...Ch. 7 - Draw the products of each SN1 reaction and...Ch. 7 - Prob. 62PCh. 7 - Prob. 63PCh. 7 - Draw a stepwise, detailed mechanism for the...Ch. 7 - When a single compound contains both a nucleophile...Ch. 7 - Prob. 69PCh. 7 - Prob. 70PCh. 7 - Draw a stepwise, detailed mechanism f or the...Ch. 7 - Prob. 72PCh. 7 - Prob. 78P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Draw a Lewis structure for each of the following species: a. H2CO3 b. CO32 c. CH2O d. CO2
Essential Organic Chemistry (3rd Edition)
Classify each example of molecular art as a pure element, a pure compound, or a mixture.
General, Organic, and Biological Chemistry - 4th edition
For each of the following 2-dimensional shapes, determine the highest order rotation axis of symmetry.
Inorganic Chemistry
What is the pH range for acidic solutions? For basic solutions?
EBK INTRODUCTION TO CHEMISTRY
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Under certain reaction conditions, 2,3-dibromobutane reacts with twoequivalents of base to give three products, each of which contains twonew π bonds. Product A has two sp hybridized carbon atoms, product Bhas one sp hybridized carbon atom, and product C has none. What arethe structures of A, B, and C?arrow_forwardDraw the structure of each alkene of molecular formula C7H14 that has a tetrasubstituted double bondarrow_forwardA is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. (a) Label each alkene that exhibits stereoisomerism as E or Z. (b) Draw a stereoisomer of A that has all Z double bonds.arrow_forward
- Draw the organic product(s) formed upon the addition of HBr to (a) 2-methyl-2-pentene, (b) trans-2-hexene, and (c) 4-methylcyclohexene. How many regioisomers can be formed in each case?arrow_forwardThe shrub ma huang (Section 5.4A) contains two biologically activestereoisomers—ephedrine and pseudoephedrine—with two stereogeniccenters as shown in the given structure. Ephedrine is one component ofa once-popular combination drug used by body builders to increaseenergy and alertness, whereas pseudoephedrine is a nasaldecongestant.a.) Draw the structure of naturally occurring (−)-ephedrine, which has the1R,2S configuration.b.) Draw the structure of naturally occurring (+)-pseudoephedrine, whichhas the 1S,2S configuration.c.) How are ephedrine and pseudoephedrine related?d.) Draw all other stereoisomers of (−)-ephedrine and (+) pseudoephedrine, and give the R,S designation for all stereogeniccenters.e.) How is each compound drawn in part (d) related to (−)-ephedrine?arrow_forwardDraw the constitutional isomer formed when the attached alkenes are treated with each set of reagents: [1] H2O, H2SO4; or [2] BH3 followed by H2O2, −OH.arrow_forward
- Draw the organic products formed when cyclopentene is treated withfollowing reagent. CH3CO3Harrow_forwardWhat product is formed when benzene is treated with each organic halide in the presence of AlCl3?arrow_forwardCan an aldehyde have molecular formula C 5H 12O? Explain why or why not.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License