
Chemistry for Engineering Students
4th Edition
ISBN: 9781337398909
Author: Lawrence S. Brown, Tom Holme
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 7, Problem 7.86PAE
7.86 Nitrogen triiodide,
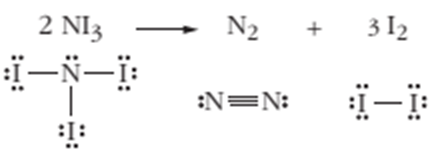
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 7 Solutions
Chemistry for Engineering Students
Ch. 7 - List some factors influencing the biocompatibility...Ch. 7 - • use electron configurations to explain why...Ch. 7 - • describe die energy changes in the formation of...Ch. 7 - • define electronegativity and state how...Ch. 7 - • identify or predict polar, nonpolar, and ionic...Ch. 7 - • write Lewis electron structures for molecules or...Ch. 7 - • describe chemical bonding using a model based on...Ch. 7 - • explain how hybridization reconciles observed...Ch. 7 - • predict the geometry of a molecule from its,...Ch. 7 - • use models (real or software) to help visualize...
Ch. 7 - • explain the formation of multiple bonds in terms...Ch. 7 - • identify sigma and pi bonds in a molecule and...Ch. 7 - Define the term biocompatibility.Ch. 7 - List some properties associated with biomaterials...Ch. 7 - Prob. 7.3PAECh. 7 - Prob. 7.4PAECh. 7 - Prob. 7.5PAECh. 7 - Prob. 7.6PAECh. 7 - Why is the ion not found in nature?Ch. 7 - Why do nonmetals tend to form anions rather than...Ch. 7 - Prob. 7.9PAECh. 7 - 7.10 Arrange the members of each of the following...Ch. 7 - 7.11 Arrange the following sets of anions in order...Ch. 7 - 7.12 Which pair will form a compound with the...Ch. 7 - 7.13 Figure 7-2 depicts the interactions of an ion...Ch. 7 - 7.14 Describe the difference between a covalent...Ch. 7 - 7.15 Covalently bonded compounds tend to have much...Ch. 7 - Prob. 7.16PAECh. 7 - 7.17 Coulombic forces are often used to explain...Ch. 7 - 7.18 In terms of the strengths of the covalent...Ch. 7 - 7.19 If the formation of chemical bonds always...Ch. 7 - 7.20 Draw the Lewis dot symbol for each of the...Ch. 7 - 7.21 Theoretical models for the structure of...Ch. 7 - 7.22 Use Lewis dot symbols to explain why chlorine...Ch. 7 - 7.23 Define the term lone pair.Ch. 7 - 7.24 How many electrons are shared between two...Ch. 7 - 7.25 How does the bond energy of a double bond...Ch. 7 - 7.26 How is electronegativity defined?Ch. 7 - 7.27 Distinguish between electron affinity and...Ch. 7 - 7.28 Certain elements in the periodic table shown...Ch. 7 - 7.29 When two atoms with different...Ch. 7 - 7.30 The bond in HF is said to be polar, with the...Ch. 7 - 7.31 Why is a bond between two atoms with...Ch. 7 - Prob. 7.32PAECh. 7 - 7.33 In each group of three bonds, which bond is...Ch. 7 - Prob. 7.34PAECh. 7 - 7.35 Which one of the following contains botb...Ch. 7 - Prob. 7.36PAECh. 7 - 7.37 Draw the Lewis structure for each of the...Ch. 7 - 7.38 Draw a Lewis structure for each of the...Ch. 7 - Prob. 7.39PAECh. 7 - 7.40 Why is it impossible for hydrogen to be the...Ch. 7 - Prob. 7.41PAECh. 7 - 7.42 Draw resonance structure for (a) (b) and (c)Ch. 7 - Prob. 7.43PAECh. 7 - Prob. 7.44PAECh. 7 - Prob. 7.45PAECh. 7 - 7.46 Consider the nitrogen-oxygen bond lengths in...Ch. 7 - 7.47 Which of the species listed has a Lewis...Ch. 7 - 7.48 Identify what is incorrect in the Lewis...Ch. 7 - 7.49 Identify what is incorrect in the Lewis...Ch. 7 - 7.50 Chemical species are said to be isoelectronic...Ch. 7 - 7.51 Explain the concept of wave interference in...Ch. 7 - 7.52 How does orbital overlap explain the buildup...Ch. 7 - 7.53 How do sigma and pi bonds differ? How are...Ch. 7 - 7.54 CO , CO2 , CH3OH , and CO32 , all contain...Ch. 7 - 7.55 Draw the Lewis dot structure of the following...Ch. 7 - 7.56 Draw the Lewis dot structures of the...Ch. 7 - 7.57 What observation about molecules compels us...Ch. 7 - Prob. 7.58PAECh. 7 - 7.59 What type of hybrid orbital is generated by...Ch. 7 - 7.60 What type of hybridization would be expected...Ch. 7 - 7.61 What hybrid orbitals would be expected for...Ch. 7 - 7.62 What type of hybridization would you expect...Ch. 7 - 7.63 What physical concept forms the premise of...Ch. 7 - 7.64 Predict the geometry of the following...Ch. 7 - Prob. 7.65PAECh. 7 - Prob. 7.66PAECh. 7 - Prob. 7.67PAECh. 7 - 7.68 Give approximate values for the indicated...Ch. 7 - 7.69 Propene has the chemical formula Describe the...Ch. 7 - Prob. 7.70PAECh. 7 - Prob. 7.71PAECh. 7 - 7.72 How does an MSN differ from amorphous silica...Ch. 7 - Prob. 7.73PAECh. 7 - 7.74 In a lattice, a positive ion is often...Ch. 7 - 7.75 Use the concept of lattice energy to...Ch. 7 - Prob. 7.76PAECh. 7 - Prob. 7.77PAECh. 7 - Prob. 7.78PAECh. 7 - Prob. 7.79PAECh. 7 - Prob. 7.80PAECh. 7 - Prob. 7.81PAECh. 7 - Prob. 7.82PAECh. 7 - Prob. 7.83PAECh. 7 - 7.84 Which of the following molecules is least...Ch. 7 - 7.85 Consider the molecule whose structure is...Ch. 7 - 7.86 Nitrogen triiodide, NI3(s) , is unstable and...Ch. 7 - 7.87 Nitrogen is capable of forming single,...Ch. 7 - 7.88 The N5+ cation has been synthesized and...Ch. 7 - Prob. 7.89PAECh. 7 - Prob. 7.90PAECh. 7 - 7.91 A Lewis structure for the oxalate ion is...Ch. 7 - Prob. 7.92PAECh. 7 - 7.93 An unknown metal M forms a chloride with the...Ch. 7 - Prob. 7.94PAECh. 7 - Prob. 7.95PAECh. 7 - 7.96 Consider the hydrocarbons whose structures...Ch. 7 - 7.97 Consider the structure shown below for as...Ch. 7 - Prob. 7.98PAECh. 7 - Prob. 7.99PAECh. 7 - Prob. 7.100PAECh. 7 - 7.101 Lead selenide nanocrystals may provide a...Ch. 7 - Prob. 7.102PAECh. 7 - Prob. 7.103PAECh. 7 - 7.104 Hydrogen azide, HN3 , is a liquid that...Ch. 7 - Prob. 7.105PAECh. 7 - Prob. 7.106PAECh. 7 - 7.107 How do the Lewis symbols for C, Si, and Ge...Ch. 7 - Prob. 7.108PAECh. 7 - Prob. 7.109PAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the reactions of silver metal, Ag(s), with each of the halogens: fluorine, F2(g), chlorine, Cl2(g), and bromine, Br2(l). What chapter data could you use to decide which reaction is most exothermic? Which reaction is that?arrow_forwardA commercial process for preparing ethanol (ethyl alcohol), C2H5OH, consists of passing ethylene gas. C2H4, and steam over an acid catalyst (to speed up the reaction). The gas-phase reaction is Use bond enthalpies (Table 9.5) to estimate the enthalpy change for this reaction when 37.0 g of ethyl alcohol is produced.arrow_forwardHydrazine, N2H4, is a colorless liquid used as a rocket fuel. What is the enthalpy change for the process in which hydrazine is formed from its elements? N2(g)+2H2(g)N2H4(l) Use the following reactions and enthalpy changes: N2H4(l)+O2(g)N2(g)+2H2O(l);H=622.2kJH2(g)+12O2(g)H2O(l);H=285.8kJarrow_forward
- For the reactions of molecular hydrogen with fluorine and with chlorine: (a) Calculate the enthalpy change for breaking all the bonds in the reactants. (b) Calculate the enthalpy change for forming all the bonds in the products. (c) From the results in parts (a) and (b), calculate the enthalpy change for the reaction. (d) Which reaction is most exothermic?arrow_forwardHydrogen, H2, is prepared by steam reforming, in which hydrocarbons are reacted with steam. For CH4, CH4(g)+H2O(g)CO(g)+3H2(g) Calculate the enthalpy change H for this reaction, using standard enthalpies of formation.arrow_forwardThe reaction of quicklime, CaO, with water produces slaked lime, Ca(OH)2, which is widely used in the construction industry to make mortar and plaster. The reaction of quicklime and water is highly exothermic: CaO(s)+H2O(l)Ca(OH)2(s)H=350kJmol1 (a) What is the enthalpy of reaction per gram of quicklime that reacts?. (b) How much heat, in kilojoules, is associated with the production of 1 ton of slaked lime?arrow_forward
- The standard enthalpies of formation for S(g), F(g), SF4(g), and SF6(g) are + 278.8, + 79.0, 775, and 1209 kJ/mol, respectively. a. Use these data to estimate the energy of an SF bond. b. Compare your calculated value to the value given in Table 8.5. What conclusions can you draw? c. Why are the Ht0 values for S(g) and F(g) not equal to zero, since sulfur and fluorine are elements?arrow_forward(a) Calculate the enthalpy change, rH, for the formation of 1.00 mol of strontium carbonate (the material that gives the red color in fireworks) from its elements. Sr(s) + C(s) + 32, O2(g) SrCO3(s) The experimental information available is Sr(s) + O2(g) SrO(s) fH = 592 kJ/mol-rxn SrO(s) + CO2(g) SrCO3(s) rH = 234 kJ/mol-rxn C(graphite) + O2(g) CO2(g) fH = 394 kJ/mol-rxn (b) Draw an energy level diagram relating the energy quantities in this problem.arrow_forwardChloromethane, CH3Cl, a compound found throughout the environment, is formed in the reaction of chlorine atoms with methane. CH4(g) + 2 Cl(g) CH3Cl(g) + HCl(g) (a) Calculate the enthalpy change for the reaction of CH4(g) and CI atoms to give CH3CI(g) and HCl(g). Is the reaction exo- or endothermic? (b) Draw an energy level diagram that shows how the various enthalpies in this problem are related.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Periodic Properties of Elements | Chemistry | IIT-JEE | NEET | CBSE | Misostudy; Author: Misostudy;https://www.youtube.com/watch?v=L26rRWz4_AI;License: Standard YouTube License, CC-BY
Periodic Trends: Electronegativity, Ionization Energy, Atomic Radius - TUTOR HOTLINE; Author: Melissa Maribel;https://www.youtube.com/watch?v=0h8q1GIQ-H4;License: Standard YouTube License, CC-BY