
Concept explainers
(a)
Interpretation:
The difficulty of that would come on models of the cis and trans isomers of
Concept introduction:
The organic compounds which consist of two cyclic rings fused together are known as bicyclic compounds. Fused rings, bridged rings, and spiro rings are the examples of bicyclic compounds. The stability of bicyclic compounds depends on the ring strain.
Answer to Problem 7.21P
The cis isomers of
Explanation of Solution
The structure of cis and trans isomers of
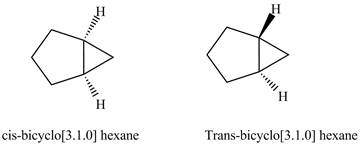
Figure 1
The formation of trans isomers of
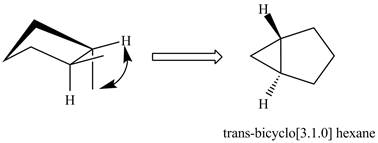
Figure 2
In the formation of trans isomers of
The formation of cis isomers of
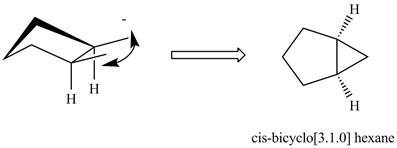
Figure 3
In the formation of cis isomers of
It is easier to form a three-membered ring by the methyl at the cis position and the methyl groups at trans position. Therefore, cis isomers of
(b)
Interpretation:
The difficulty of that would come on models of
Concept introduction:
The organic compounds which consist of two cyclic rings fused together are known as bicyclic compounds. Fused rings, bridged rings and spiro rings are the examples of bicyclic compounds. The stability of bicyclic compounds depends on the ring strain.
Answer to Problem 7.21P
The compound
Explanation of Solution
The structure of
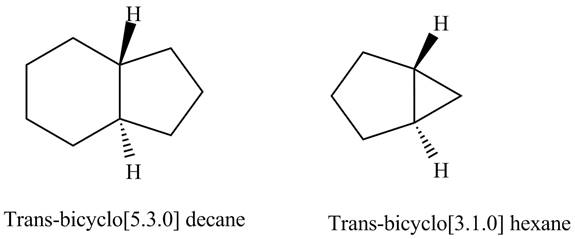
Figure 4
The formation of
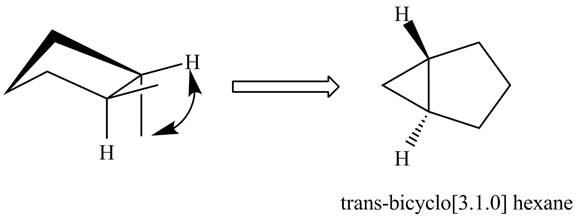
Figure 2
In the formation of
The formation of
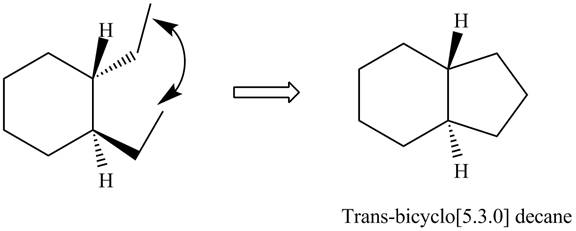
Figure 5
In the formation of
The methyl groups in case of
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry
- this is an organic chemistry question please answer accordindly!! please post the solution draw the figures and post, answer the question in a very simple and straight forward manner thanks!!!!! please answer EACH part till the end and dont just provide wordy explanations wherever asked for structures or diagrams, please draw them on a paper and post clearly!! answer the full question with all details EACH PART CLEARLY please thanks!! im reposting this kindly solve all parts and draw it not just word explanations!!arrow_forwardPlease correct answer and don't used hand raitingarrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Select to Edit Arrows H H Select to Add Arrows > H CFCI: Select to Edit Arrows H Select to Edit Arrowsarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

