
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 38P
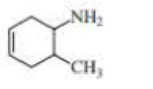
What is each compound’s systematic name?
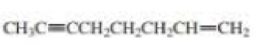
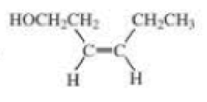
- a. CH3CH2C≡CCH2CH2C≡CH
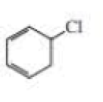
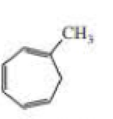
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
А
H.
OH
C
CH,CH3
D
E
NH
CH2CH3
CH2CH,
CH2
H3C
H3C
COOH
Reagents
e. PB13
f. NaCN then H3O*
g. NBS, CCI4
h. CrO3, H2SO4
а.
HBr
b. Mg, ether
c. CO2, ether then H3O*
d. BH3, THF then H2O2, OH"
Indicate the product obtained by reacting A and B.
A
||
H3C-S-CH₂ Na*
||
CH 3
B
CH2.
C.
CH3
H3C
CH3
Chapter 7 Solutions
Organic Chemistry
Ch. 7.1 - What is the molecular formula for a monocyclic...Ch. 7.1 - Prob. 2PCh. 7.1 - Draw the structure and give the common and...Ch. 7.1 - Prob. 4PCh. 7.1 - Name the following:Ch. 7.2 - Prob. 6PCh. 7.2 - Name the following:Ch. 7.3 - Prob. 8PCh. 7.3 - Why does cis-2-butene have a higher boiling point...Ch. 7.4 - What orbitals are used to form the carbon-carbon ...
Ch. 7.6 - Prob. 12PCh. 7.6 - Prob. 13PCh. 7.7 - Prob. 14PCh. 7.7 - Which alkyne should be used for the synthesis of...Ch. 7.7 - Prob. 16PCh. 7.8 - Prob. 17PCh. 7.8 - Only one alkyne forms an aldehyde when it...Ch. 7.9 - Describe the alkyne you should start with and the...Ch. 7.9 - Prob. 20PCh. 7.10 - Prob. 21PCh. 7.10 - Prob. 22PCh. 7.10 - Rank the following from strongest base to weakest...Ch. 7.12 - Prob. 26PCh. 7 - What is the major product obtained from the...Ch. 7 - Draw a condensed structure for each of the...Ch. 7 - A student was given the structural formula of...Ch. 7 - Prob. 30PCh. 7 - What is each compounds systematic name?Ch. 7 - What reagents should be used to carry out the...Ch. 7 - Prob. 33PCh. 7 - Prob. 34PCh. 7 - Prob. 35PCh. 7 - Prob. 36PCh. 7 - What is the major product of the reaction of 1 mol...Ch. 7 - What is each compounds systematic name? a....Ch. 7 - What is the molecular formula of a hydrocarbon...Ch. 7 - Answer Problem 39, parts a-b, using 2-butyne as...Ch. 7 - Prob. 41PCh. 7 - a. Starting with 3-methyl 1-butyne, how can you...Ch. 7 - Prob. 43PCh. 7 - Which of the following pairs are keto-enol...Ch. 7 - Prob. 45PCh. 7 - Do the equilibria of the following acid-base...Ch. 7 - What stereoisomers are obtained when 2-butyne...Ch. 7 - Draw the keto tautomer for each of the following:Ch. 7 - Show how each of the following compounds can be...Ch. 7 - A chemist is planning to synthesize 3-octyne by...Ch. 7 - Prob. 51PCh. 7 - What stereoisomers are obtained from the following...Ch. 7 - Prob. 53PCh. 7 - Prob. 54PCh. 7 - Prob. 55PCh. 7 - Prob. 56PCh. 7 - Prob. 57P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Reagents a. C6H5CHO b. NaOH, ethanol h. BrCH2CH=CH2 i. Na* OEt, ethanol j. Br2, H* k. K* t-BuO c. Pyrrolidine, cat. H* d. H2C=CHCN e. H3O* f. I. CH2(CO2ET)2 -CH2CH2CN LDA m. heat g. ELOC(=0)CO2ET Select reagents from the table to synthesize this compound from cyclopentanone. Enter the letters of the chosen reagents, in the order that you wish to use them, without spaces or punctuation (i.e. geda).arrow_forwardCONH₂ CH3 CH3 H₂C=CHCHCH₂CO₂CH3 1. LiAlH4 2. H₂O 1. LiAlH4 2. H3O+arrow_forward12) Which structure is different from the compound (I) ? CH₂ CH₂- H H a H CH(CH3)2 H. H CH₂ CH₂ H b -CH(CH₂)2 CH₂ CH₂- H H C H CH(CH₂)2 H7 CH₂ CH₂ CH(CH3)2 darrow_forward
- 4. Use the IUPAC Nomenclature System to name each of the following compounds: a. b. CH,CCH₂CH, 0 HCCHCH₂CH₂ CI O -C-C-CH₂ Br CHỊCH,CH,CH, CH,CCH₂CH₂CH CH, CI CH₂CHCH₂CH Br CH, Ô CH,CCH.CCH.CH.CH, OH 0 CH₂ CH,CCH₂CH.CH,CHCH,C-H H,CHCH.CH CH₂arrow_forwardNHČCH: CH,NH, NH2arrow_forwardDraw the products formed when CH3CH2C ≡ C−Na+reacts with each compound. a. CH3CH2CH2Brb. (CH3)2CHCH2CH2Clc. (CH3CH2)3CCld. BrCH2CH2CH2CH2OHe. ethylene oxide followed by H2Of. propene oxide followed by H2Oarrow_forward
- Name each alkyne. a. CH=C-CH-CH3 CH3 b. CH3-C=C-CH-CH-CH,-CH, CH3 CH3 c. CHEC-C-CH,-CH3 CH2 ČH, CH3 d. CH;-C=C-CH-C-CH, CH2 CH3 ČH3arrow_forward1. Provide proper IUPAC names for the following compounds. a. CH3 | CH3C CCHCH2CH2CH3 CH3 Br b. H₂C=CH-C-CH-CC-H | CH3 C.arrow_forwardThis compound has the common name acetone. What is its proper systematic name? O C-C- C-C сarrow_forward
- 6. What is the product? CH3 осно A. C. A and B A. A B. B C. C CH3 B. DBN CH₂arrow_forwardWhich structure below shows the same compound as the following? NH₂ CH₂ CO₂H H HNH, C CH3 CO₂H b. CO₂H H₂N H CH3 Oc. CO₂H H -NH2 CH3 NH2 CH CH3 CO₂H Tiarrow_forwardIdentify the best reagents to complete the following reaction. CI N.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY