
Concept explainers
What is the major product obtained from the reaction of each of the following compounds with excess HCl?
- a. CH3CH2C≡CH
- b. CH3CH2C≡CCH2CH3
- c. CH3CH2C≡CCH2CH2CH3
(a)
Interpretation:
The major product obtained from the reaction of given compound with excess of
Concept Introduction:
Addition of halides to alkynes: Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.
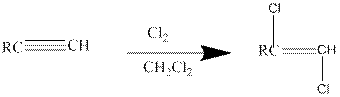
If excess halides are present, a second addition reaction also occurs.
Answer to Problem 29P
The major product obtained from the reaction of given compound with excess of

Explanation of Solution
Hydrogen halides undergo addition reaction with alkynes. Alkyne is a nucleophile and it reacts with the electrophile to give a

In this reaction of butyne with
(b)
Interpretation:
The major product obtained from the reaction of given compound with excess of
Concept Introduction:
Addition of halides to alkynes: Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.
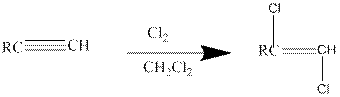
If excess halides are present, a second addition reaction also occurs.
Answer to Problem 29P
The major product obtained from the reaction of given compound with excess of

Explanation of Solution
Hydrogen halides undergo addition reaction with alkynes. Alkyne is a nucleophile and it reacts with the electrophile to give a

In this reaction of hex-3-yne with
(c)
Interpretation:
The major product obtained from the reaction of given compound with excess of
Concept Introduction:
Addition of halides to alkynes: Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.
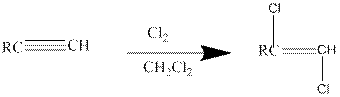
If excess halides are present, a second addition reaction also occurs.
Answer to Problem 29P
The major product obtained from the reaction of given compound with excess of
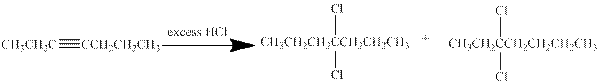
Explanation of Solution
Hydrogen halides undergo addition reaction with alkynes. Alkyne is a nucleophile and it reacts with the electrophile to give a
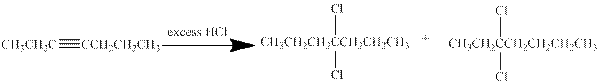
In this reaction of hept-3-yne with
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry (8th Edition)
- Which alkyl halide will react the fastest in an SN2 reaction (CH3)3CI CH3CH2Br CH3CH2I CH3I CH3Brarrow_forwardIn which compound is the halogen substituted most rapidly by aq hydroxide ions? a. CH3CH2CH2CH2Cl b. (CH3)3CI c. (CH3)3CCl d. CH3CH2CH2CH2Iarrow_forwardWhat are the correct reagents for this reaction? A.) NaBH4 and then H3O+ B.) LiAlH4/ether and then H3O+ C.) H2 with Pt/C D.) all of the abovearrow_forward
- Choose the appropriate reagent for the following reaction. A) TsCl/pyridine B) SOCl2/pyridine C) ZnCl2 D) PBr3 E) HIarrow_forwardWhich alkyl halide in each of the following pairs will react faster in an SN2 reaction with hydroxyl ion? 3.2 CH3CI or (CH3)3CCI 3.3 CH3CH=CHBr or CH2=CHCH2Br 3.4 Reaction of CN- with CH3CH(Br)CH3 or with CH3CH2CH2Brarrow_forwardWhat is the major E2 elimination product formed from each alkyl halide?arrow_forward
- What is the name of reaction 2? a Grignard reaction b Reduction c Acetal formation d Williamson ether synthesis Which of the following is product B? a Methoxycyclopentane b 1-methylcyclopentan-1-ol c 1,2-methoxycyclopentane d Cyclopentanonearrow_forwardThe reaction H2C=CHCH2Br + NaN3 followed by reaction with LiAlH4 yields CH3CH2CH2NH2 H2C=CHCH2NH2 H2C=CHCH2N3 none of the abovearrow_forward1. What type of reaction is occuring in step 3? (halogenation, hydrohalogenation, reduction, keto–enol tautomerism, dehydrohalogenation, acid-catalyzed hydration, base-catalyzed hydration) 2. Which reagent is necessary for step 3? (Br2, HBr, H2/Pt, NaNH2, H20/H2SO4/HgSO4)arrow_forward
- Pick the reactant or solvent in each part that gives the faster SN1 reaction.a. reaction of H2O with (CH3)3CCl or (CH3)3CIb. reaction of CH3OH with (CH3)3CBr or (CH3)2CHCH2Brc. reaction of CH3CH2CH(I)CH3 with CH3CH2OH in water or DMSOarrow_forwardWhat is the major product of the reaction of 2-methyl-2-butene with each of the following reagents? a. HBr b. HI c. Cl2/CH2Cl2 d. O3, −78 °C, followed by (CH3)2S e. H2/Pd f. MCPBA (a peroxyacid) g. H2O + H2SO4 h. Br2/CH2Cl2i. Br2/H2O j. Br2/CH3OH k. BH3 /THF, followed by H2O2, HO-, H2Oarrow_forwardWhich of the following compounds will dissolve in aqueous NaOH after undergoing reaction with Hinsberg reagent? CH3CH2NH2 (CH3)3N (CH3CH2)2NH (CH3CH2)3Narrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
