
Concept explainers
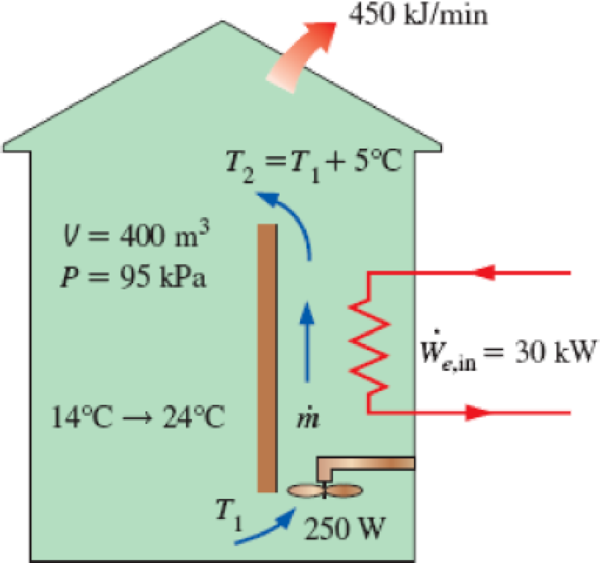
A building with an internal volume of 400 m3 is to be heated by a 30-kW electric resistance heater placed in the duct inside the building. Initially, the air in the building is at 14°C, and the local atmospheric pressure is 95 kPa. The building is losing heat to the surroundings at a steady rate of 450 kJ/min. Air is forced to flow through the duct and the heater steadily by a 250-W fan, and it experiences a temperature rise of 5°C each time it passes through the duct, which may be assumed to be adiabatic.
- (a) How long will it take for the air inside the building to reach an average temperature of 24°C?
- (b) Determine the average mass flow rate of air through the duct.
FIGURE P5–173
(a)
The time taken to attain the building’s average temperature of
Answer to Problem 173RP
The time taken to attain the building’s average temperature of
Explanation of Solution
Consider the entire building as system and the air circulates the in the building itself. There is no leakage to the surrounding.
The air flows at steady state through one inlet and one exit system (pipe and duct flow). Hence, the inlet and exit mass flow rates are equal.
Write the energy balance equation.
Here, the heat transfer is
In this system two work inputs are involved namely, the work input to the electric heater
The Equations (I) reduced as follows.
Here, there is no mass leakage from the building to the surrounding. The mass of air circulates in the building itself. Hence, inlet and exit enthalpies are neglected.
The change in internal energy is expresses as follow.
Here, the specific heat at constant volume is
Neglect the inlet and exit enthalpies and substitute
Equation (II).
Express the Equation (III) with respect to change of time and rearrange it to obtain
Write the formula for mass of air
The mass flow rate
Here, the change in time or time interval is
Refer Table A-1, “Molar mass, gas constant, and critical-point properties”.
The gas constant of air
Refer Table A-2, “Ideal-gas specific heats of various common gases”.
The specific heat at constant volume
Conclusion:
Substitute
Substitute
Substitute
Thus, the time taken to attain the building’s average temperature of
(b)
The average mass flow rate of air through the duct.
Answer to Problem 173RP
The average mass flow rate of air through the duct is
Explanation of Solution
Consider the heating duct with fan and heater only as the system. The air passes through in it steadily.
The system is at steady state. Hence, the rate of change in net energy of the system becomes zero.
The heating duct is an adiabatic duct. Hence, there is no heat loss.
The Equations (II) reduced as follows.
Express the Equation (VII) with respect to change of time as follows.
The change in enthalpy is expresses as follow.
Here, the specific heat at constant pressure is
Substitute
Refer Table A-2, “Ideal-gas specific heats of various common gases”.
The specific heat at constant pressure
Conclusion:
It is given that the temperature rise is
Substitute
Thus, The average mass flow rate of air through the duct is
Want to see more full solutions like this?
Chapter 5 Solutions
Thermodynamics: An Engineering Approach
- A hair dryer is basically a duct in which a few layers of electric resistors are placed. A small fan pulls the air in and forces it through the resistors where it is heated. Air enters a 1200-W hair dryer at 100 kPa and 22°C and leaves at 47°C. The cross-sectional area of the hair dryer at the exit is 60 cm2 . Neglecting the power consumed by the fan and the heat losses through the walls of the hair dryer, determine the velocity of the air at the exit.arrow_forwardWater jacketed compressor compresses 5 m³/min of air having an initial specific volume of 0.8m³/kg. During the compression, the enthalpy of air increases by 83.7 kJ/kg and heat is transferred to the cooling water at the rate of 251.1 kJ/min. Determine the work in hp to drive the compressor. 1 hp = 0.746kW = 0.746kJ/sarrow_forwardIf the container is placed in a colder environment, how will it affect the actual heat transfer and the irreversibility of the system?arrow_forward
- In large compressors, the gas is often cooled while being compressed to reduce the power consumed by the compressor. Explain how cooling the gas during a compression process reduces the power consumption.arrow_forwardConsider an adiabatic turbine operating steadily. Does the work output of the turbine have to be equal to the decrease in the energy of the steam flowing through it?arrow_forwardA piston–cylinder device initially contains 1.4 kg of refrigerant-134a at 100 kPa and 20°C. Heat is now transferred to the refrigerant from a source at 150°C, and the piston, which is resting on a set of stops, starts moving when the pressure inside reaches 120 kPa. Heat transfer continues until the temperature reaches 80°C. Assuming the surroundings to be at 25°C and 100 kPa, determine the work done.arrow_forward
- Consider a rigid vessel that is initially empty, connected by a valve to a feed line of water at 1 MPa and 280 degrees centigrade. The valve is then opened and the water flows slowly into the container until it reaches pressure P2 and the valve is closed. Determine the final temperature (T2) of the steam in the container in the following interval 0.45Mpa < P2< 1.75 Mpa and a heat loss of 51.6 Kj/Kgarrow_forwardDetermine the work required (in kJ/kg) for a pump that is used to increase the pressure of a liquid water stream from 100 kPa to 1500 kPa isothermally at 20 °C. The specific heat of water (C) is 0.0035 kJ/kg.K. its specific volume is 0.001031 m³/kg and its volume expansivity (B) is 250x10 Karrow_forwardWater is heated in an insulated, constant-diameter tube by a 7-kW electric resistance heater. If the water enters the heater steadily at 20°C and leaves at 75°C, determine the mass flow rate of water.arrow_forward
- A 5-ft3 rigid tank initially contains refrigerant-134a at 60 psia and 100 percent quality. The tank is connected by a valve to a supply line that carries refrigerant-134a at 140 psia and 80F. The valve is now opened, allowing the refrigerant to enter the tank, and is closed when it is observed that the tank contains only saturated liquid at 100 psia. Determine (a) the mass of the refrigerant that entered the tank, (b) the amount of heat transfer with the surroundings at 708F, and (c) the entropy generated during this process.arrow_forwardElectric heating systems used in many homes consist of a simple duct with resistance heaters. As it flows over the resistance wires, the air is heated. Consider a 25 kW electric heating system. Air enters the heating section with an air flow rate of 120 m3 / min at 100 kPa and 12 ° C. If 150 W of heat is lost from the air in the duct to the environment, determine the exit temperature of the air.arrow_forwardTwo constant-volume tanks, each filled with 30 kg of air, have temperatures of 900 K and 300 K. A heat engine placed between the two tanks extracts heat from the high-temperature tank, produces work, and rejects heat to the low-temperature tank. Determine the maximum work that can be produced by the heat engine and the final temperatures of the tanks. Assume constant specific heats at room temperature.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





