
To find:
The Bond order of N2+, O2+, C2+, and Br22- from the MO theory and predict the bond orders.
Answer to Problem 5.97QA
Solution:
1) Bond order of N2+ = 2.5
2) Bond order of O2+ = 2.5
3) Bond order of C2+ = 1.5
4) Bond order of Br22- = 0
All species with nonzero bond order may exist, therefore, N2+, O2+ and C2+ may exist.
Explanation of Solution
The Molecular orbital diagram of N2+, O2+, C2+, and Br22- are below
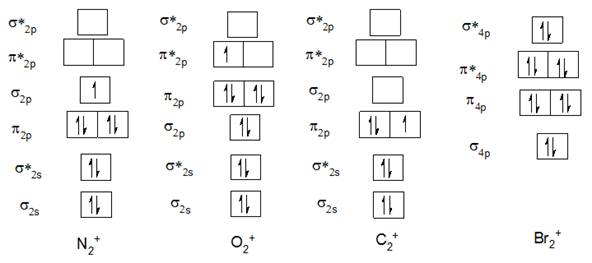
1) Bond order of N2+
:
Electronic configuration of N2+ is
2) Bond order of O2+:
Electronic configuration of O2+ is
3) Bond order of C2+:
Electronic configuration of C2+ is
4) Bond order of Br22- :
In case of Br22- after 4s there is 3d subshell which makes the orbital diagram complicated and as it is completely filled, it is going to cancel out with bonding and antibonding electrons;hence, it is neglected in this case. Valence electronic configuration of Br22- is
The bond order of Br22- is 0 because bonding and antibonding electrons are equal.
All species with nonzero bond order may exist, therefore, N2+, O2+ and C2+ may exist.
Conclusion:
The bond order of the given molecular ions is calculated from the MO diagram. It is used to check the existence of species from the bond order.
Want to see more full solutions like this?
Chapter 5 Solutions
Chemistry: An Atoms-Focused Approach
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





